Quick Navigation:
| | | |
The Amazing World of Solutes, Solvents, and Solutions
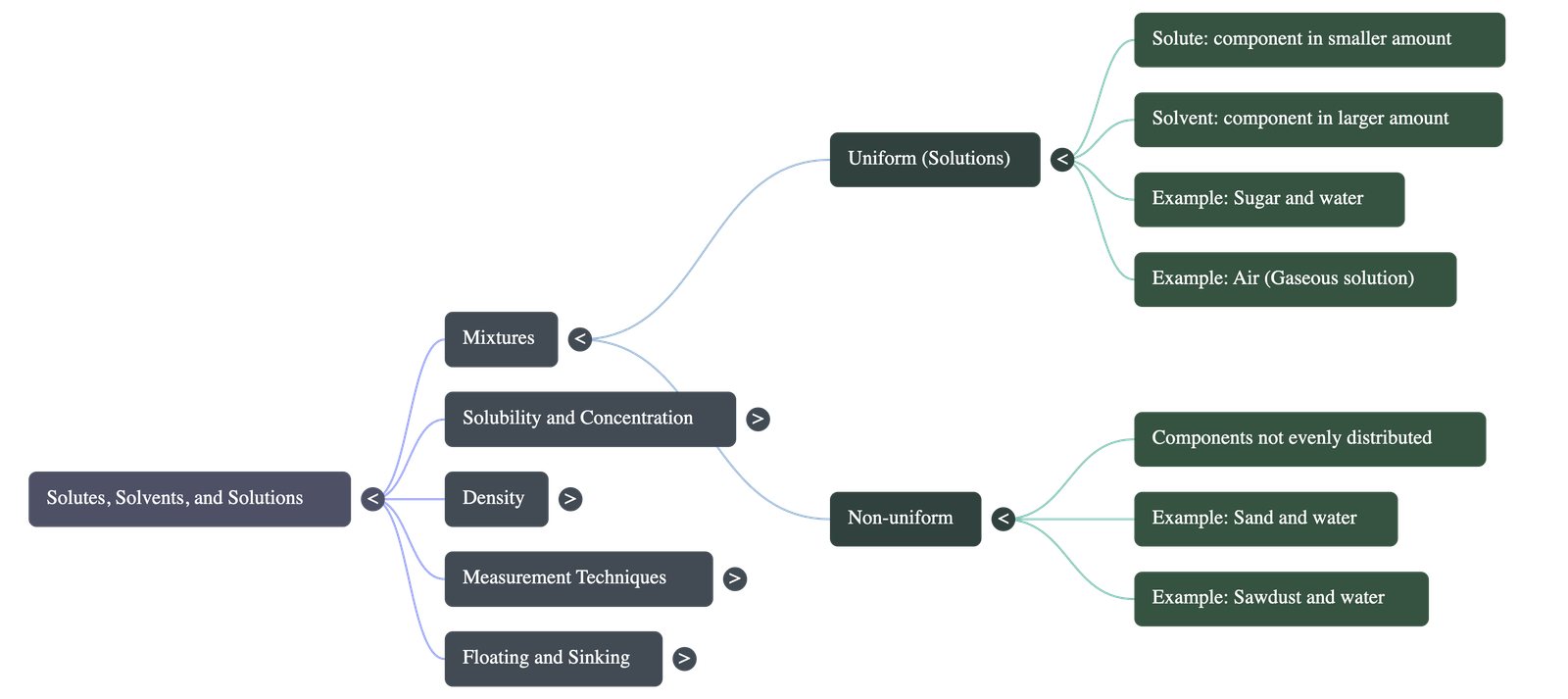
1. Types of Mixtures
- Uniform Mixtures (Solutions): Mixtures where the components are evenly distributed throughout. A common example is salt or sugar completely dissolved in water.
- Non-Uniform Mixtures: Mixtures where components are not evenly distributed, and the individual substances can often be seen (e.g., sand, chalk powder, or sawdust mixed with water).
2. Solutes, Solvents, and Solutions
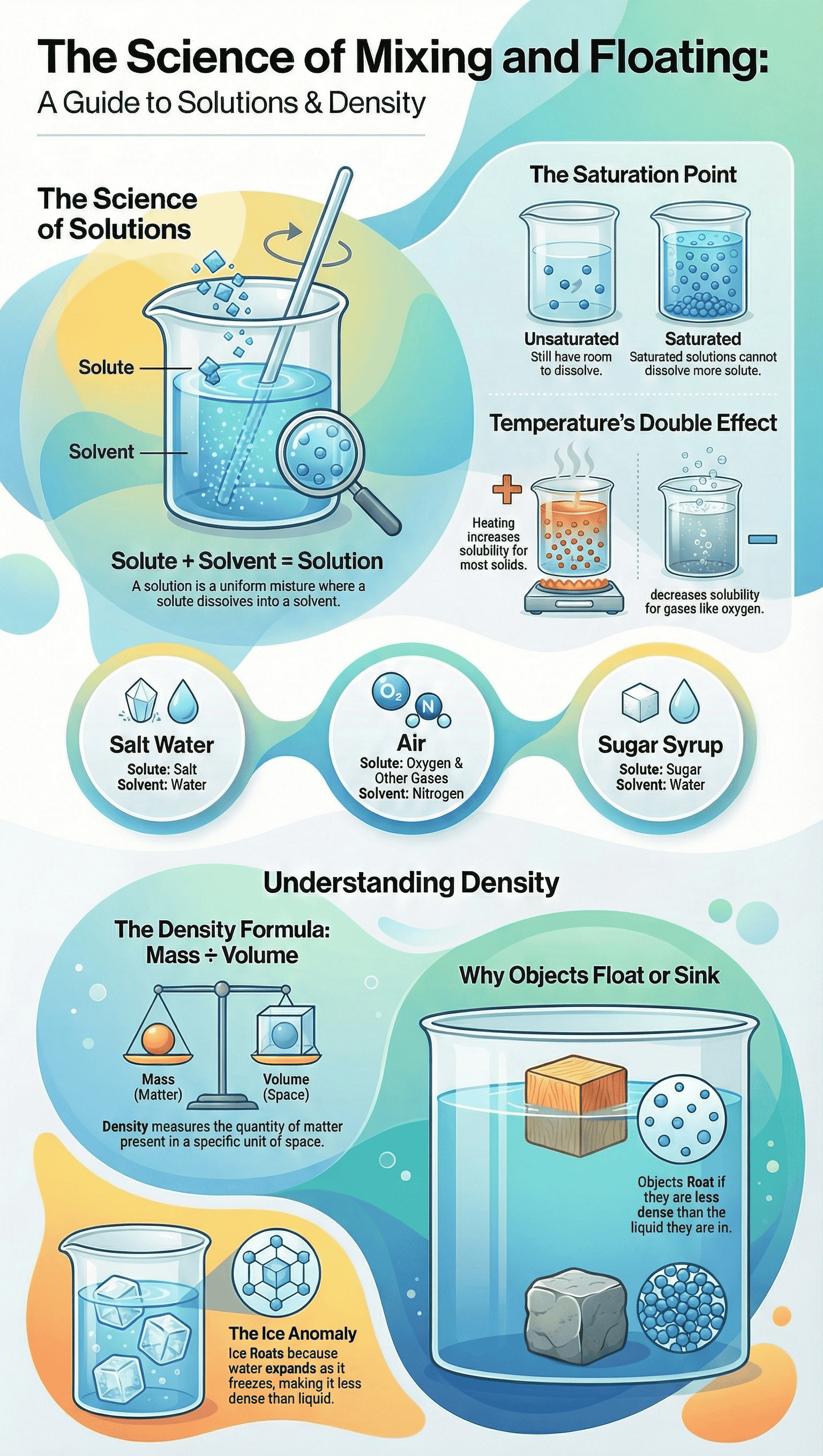
- Basic Definitions: In a uniform mixture consisting of a solid and a liquid, the solid is called the solute and the liquid is the solvent. Together, they form a solution.
- Liquid-Liquid and Gas Mixtures: When two liquids are mixed, the substance in the smaller amount is the solute, and the one in the larger amount is the solvent.
- Gaseous Solutions: Air is a solution of gases. Since Nitrogen is present in the largest quantity, it acts as the solvent, while Oxygen, Argon, Carbon dioxide, and other gases are the solutes.
- Exceptions in Proportions: In specific cases like sugar syrup (Chashni) used for sweets, the sugar (solid) may be larger in quantity than water, but water is still considered the solvent.
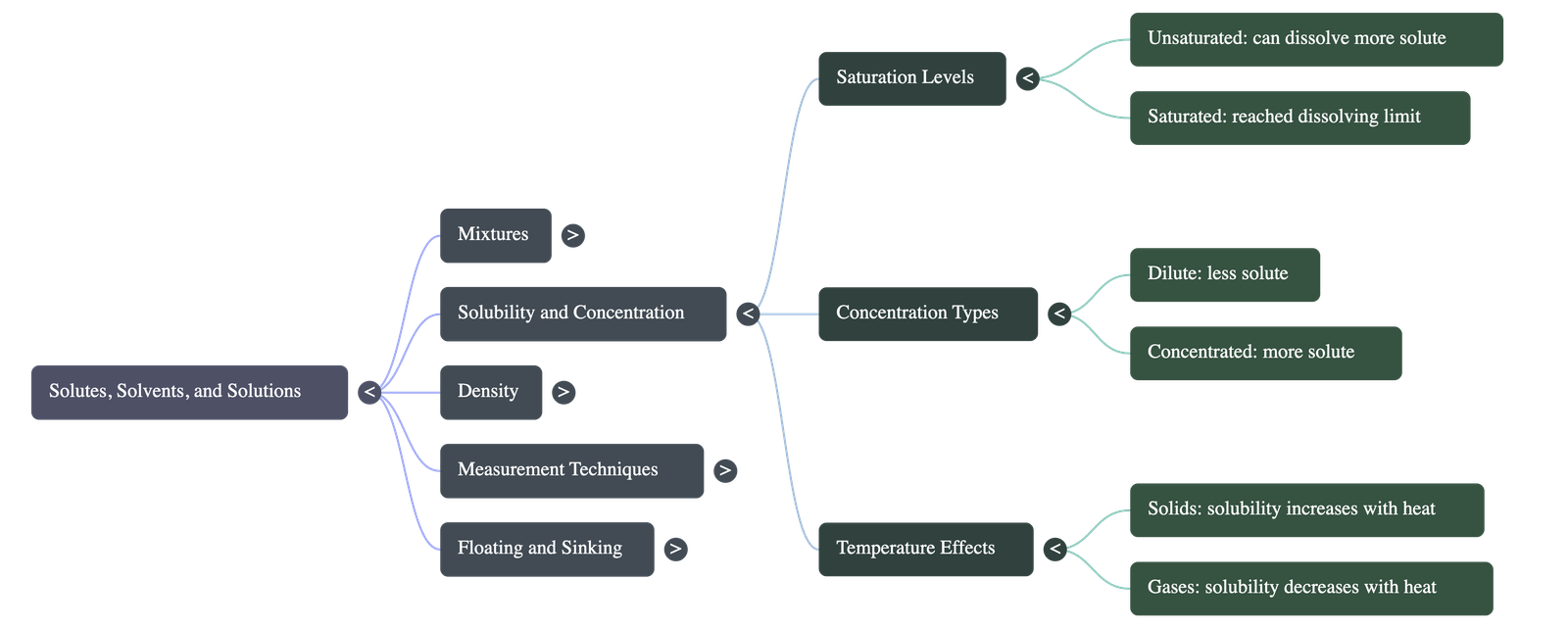
3. Solubility and Concentration
- Unsaturated vs. Saturated: An unsaturated solution can still dissolve more solute at a given temperature. A saturated solution has reached its limit; any additional solute will simply settle at the bottom.
- Concentration: Refers to the amount of solute in a fixed quantity of solution. It dictates whether a solution is dilute (less solute) or concentrated (more solute).
- Solubility: The maximum amount of solute that can dissolve in a fixed quantity of solvent at a specific temperature.
- Effect of Temperature on Solids: For most solid solutes, increasing the temperature increases their solubility. Heating a saturated solution allows it to dissolve more solute, effectively turning it back into an unsaturated solution.
- Effect of Temperature on Gases: Unlike solids, the solubility of gases generally decreases as temperature increases. For example, cold water dissolves more oxygen, which is essential for sustaining aquatic life.
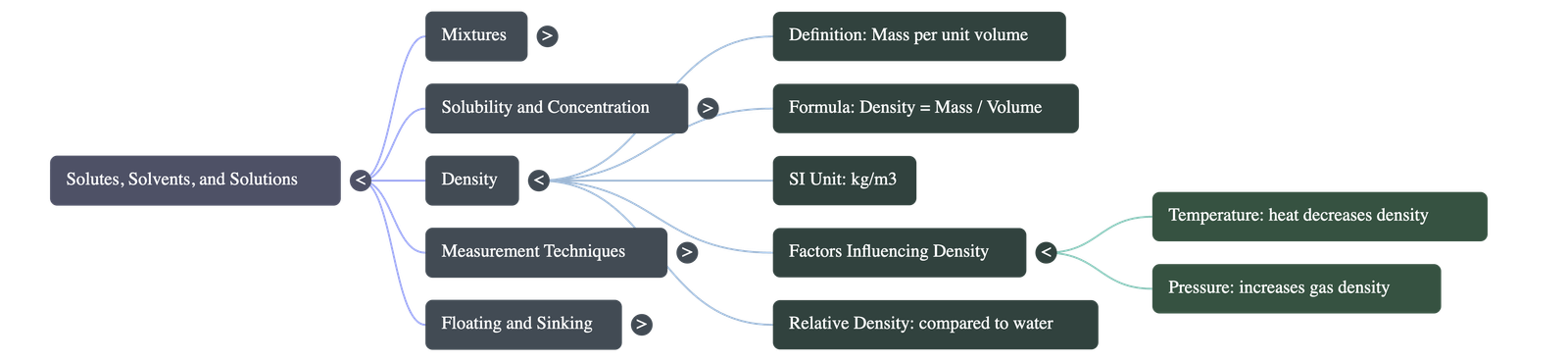
4. Understanding Density
- Definition: Density is defined as the mass present in a unit volume of a substance (Formula: Density = Mass ÷ Volume). It determines the relative "heaviness" of an object regardless of its overall shape or size.
- Units of Measurement: The standard SI unit is kg/m³. For convenience, especially with liquids, it is often expressed in g/cm³ or g/mL (where 1 mL = 1 cm³).
- Relative Density: This is a comparison of a substance's density to the density of water. It is a pure number with no units.
- Floating and Sinking: Objects that are denser than a liquid will sink, while those that are less dense (lighter) will float. For instance, oil floats on water because its density is lower.
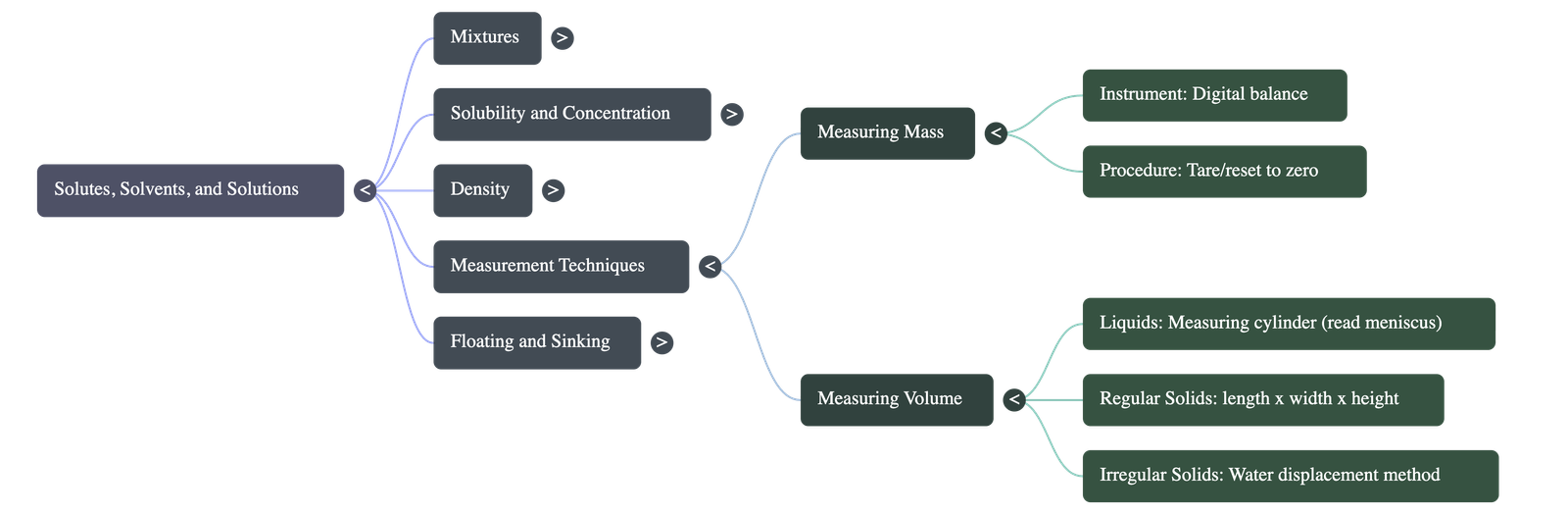
5. Measuring Mass and Volume
- Measuring Mass: Mass is the quantity of matter in an object, traditionally measured using a weighing balance. Modern setups use digital balances that can be reset to zero (using a tare button) to ignore the weight of the container.
- Measuring Volume of Liquids: Usually done using a measuring cylinder. When reading the volume, one must look at the curved surface of the liquid, known as the meniscus. Read the bottom of the meniscus for colorless liquids (like water) and the top for colored liquids.
- Measuring Volume of Solids:
- Regular Shapes: Calculated mathematically (e.g., Length × Width × Height for a cuboid).
- Irregular Shapes: Calculated using the water displacement method. The object is lowered into a measuring cylinder filled with water, and the rise in the water level corresponds to the object's volume.
6. Factors Influencing Density
- Temperature: Generally, heating a substance causes its particles to spread out (volume increases), which decreases its overall density. Cooling increases density. This principle explains why hot air balloons rise (hot air is less dense than cool air).
- The Anomaly of Water: Water is a unique exception. Its highest density is at 4 °C. As it freezes into ice at 0 °C, its particles arrange in a structure that takes up more space (expansion), meaning ice becomes less dense than liquid water. This is why ice floats, protecting underwater ecosystems during freezing temperatures.
- Pressure: Increasing pressure heavily compresses gases, significantly increasing their density. However, because liquids and solids are relatively incompressible, pressure has negligible effects on their density.
- Earth's Structure: The Earth is layered by density. The outermost crust is the lightest, and the density progressively increases deep into the inner core due to immense pressure and heat compacting the materials.
Quick Navigation:
| | | |
1 / 1
Quick Navigation:
| | | |