Quick Navigation:
| | | |
Particulate Nature of Matter
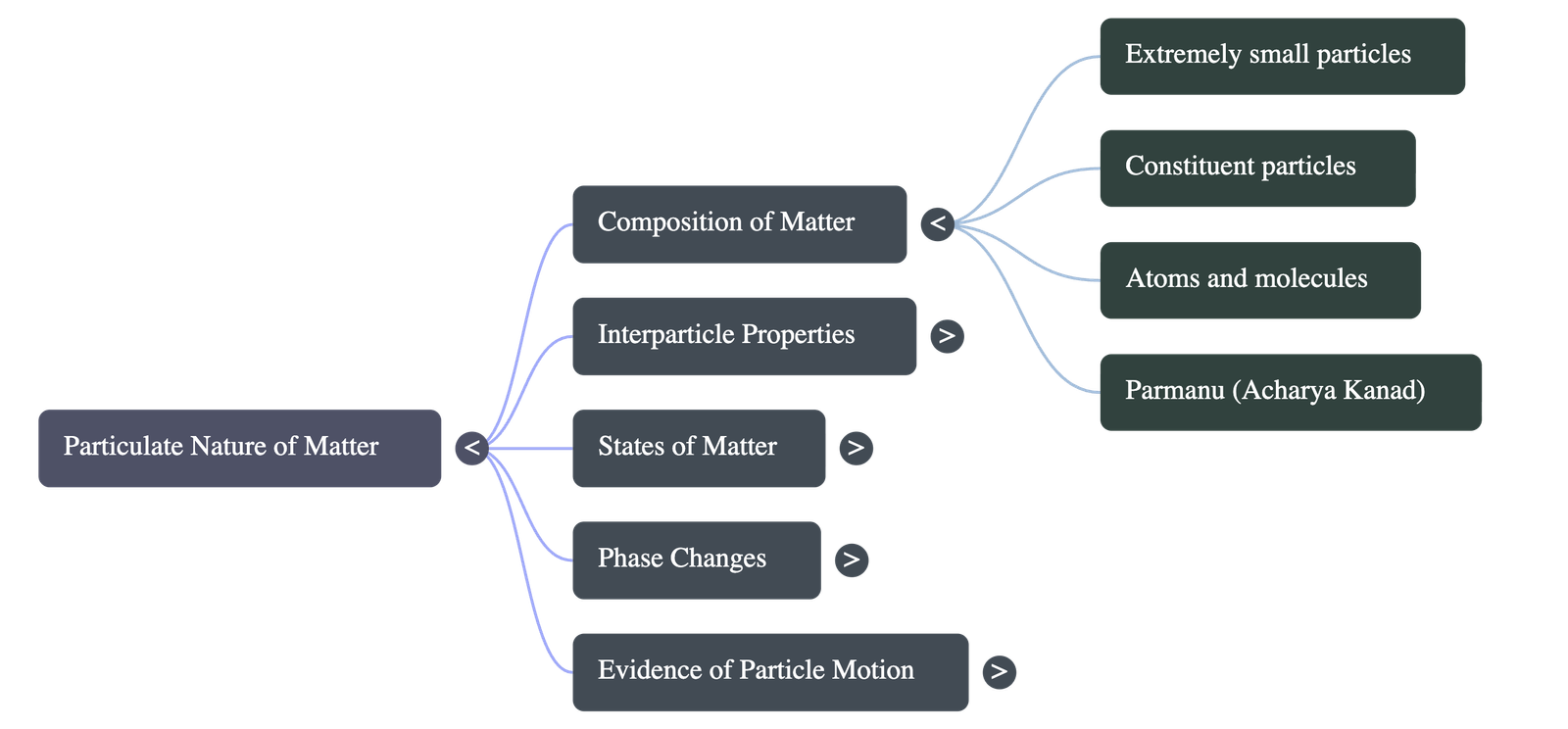
1. Composition of Matter
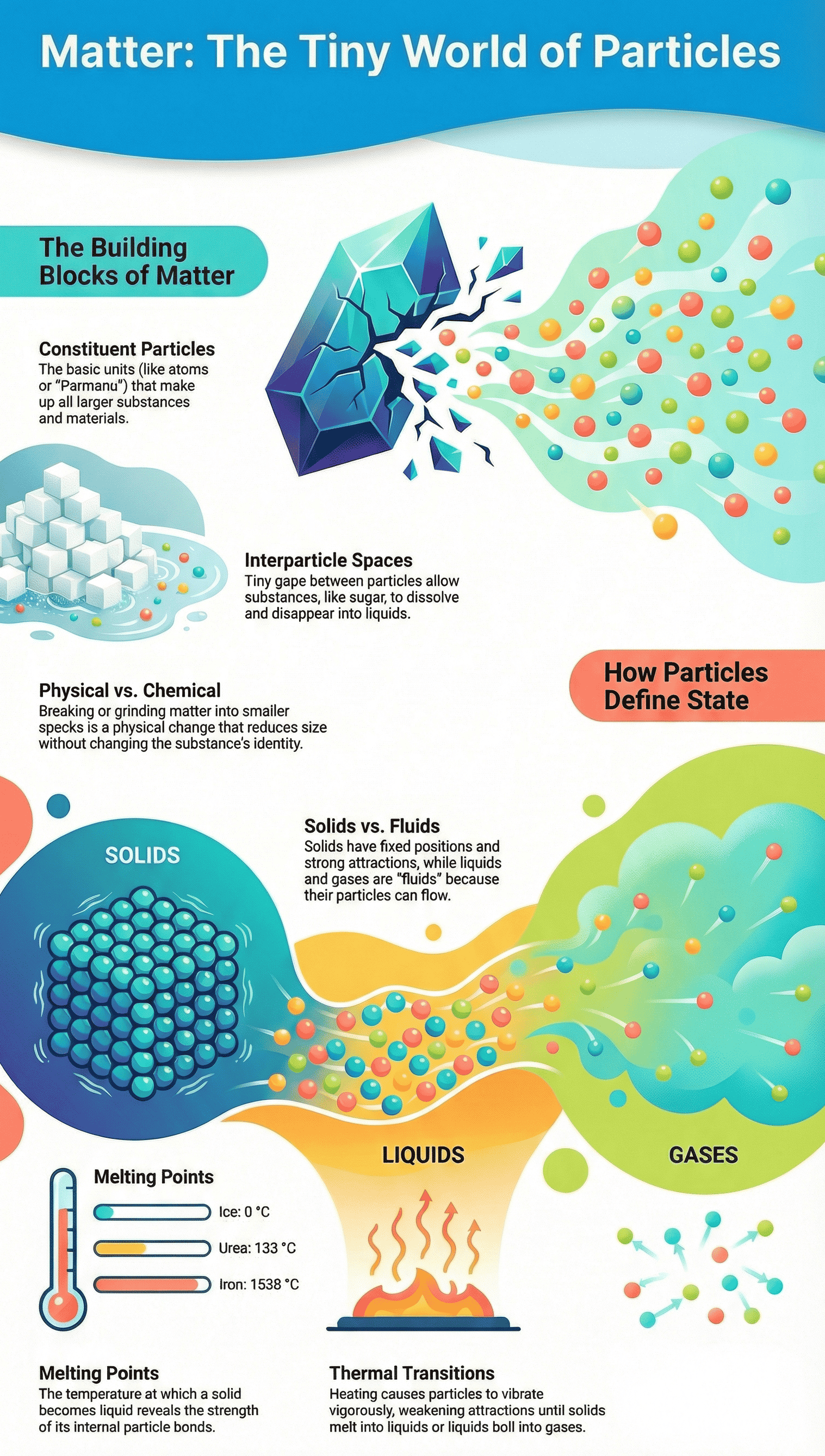
- Matter is entirely composed of an unimaginably large number of extremely small units called constituent particles, which are invisible even under ordinary microscopes.
- Breaking physical objects (like grinding chalk) or dissolving substances (like mixing sugar into water) reveals that these materials break down into these fundamental building blocks.
- The ancient Indian philosopher Acharya Kanad historically proposed this concept, referring to these tiny, indivisible, and eternal particles as Parmanu (atoms).
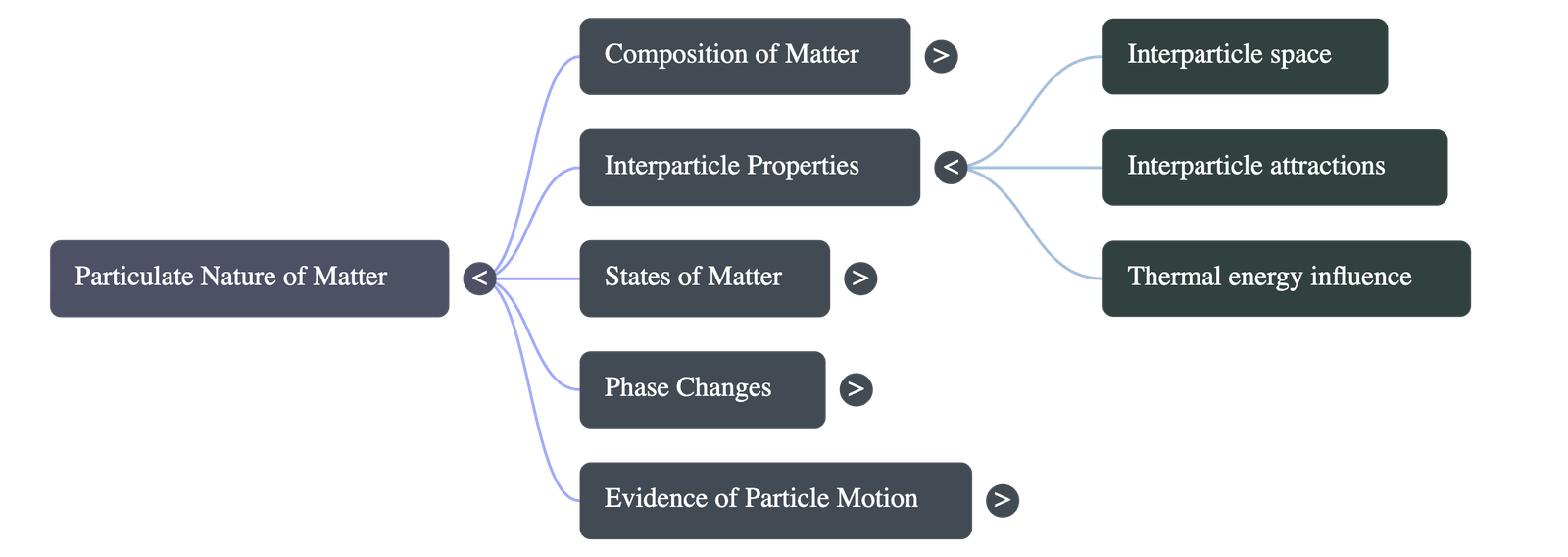
2. Interparticle Spaces and Forces
- Interparticle Spaces: There are empty spaces between the particles of matter. When a substance like sugar dissolves in water, its particles separate and occupy the invisible spaces between the water particles, which is why the overall volume does not significantly increase.
- Interparticle Attractions: Particles are held together by attractive forces. The strength of these forces depends heavily on the distance between the particles and the specific nature of the substance.
- The physical state of any substance (whether it is a solid, liquid, or gas) is ultimately dictated by the strength of these attractive forces and the spacing between its particles.
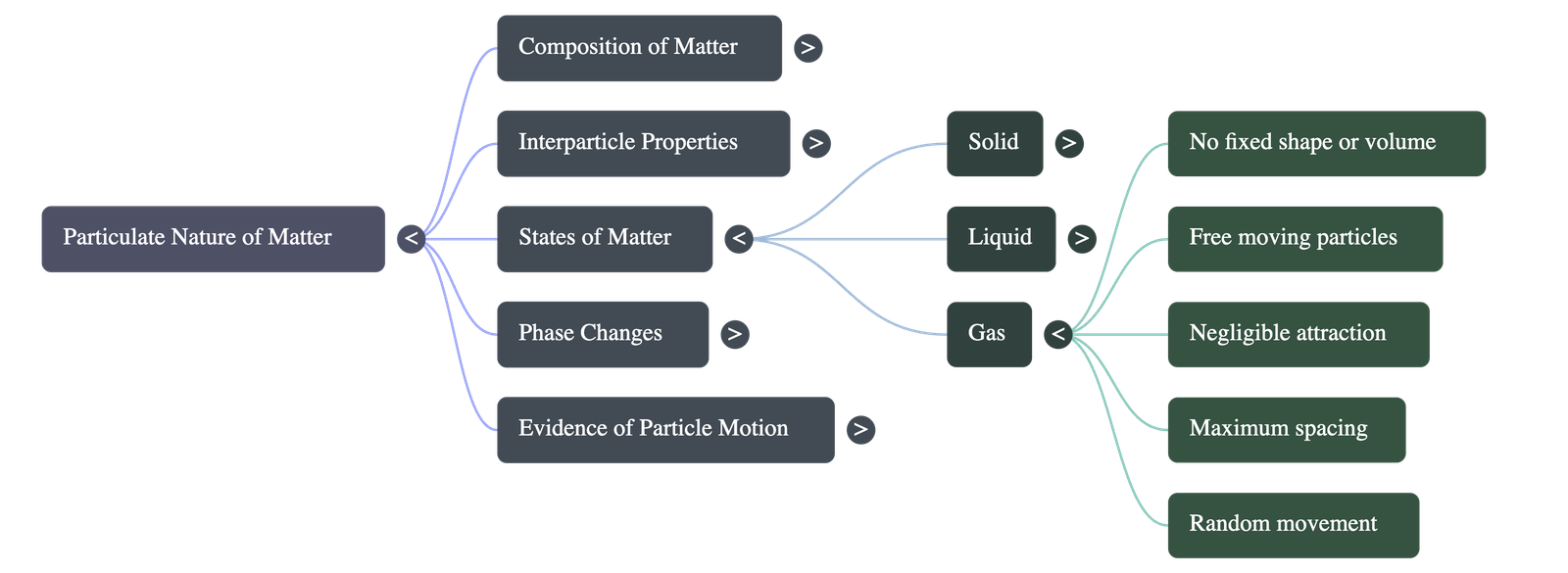
3. The Three States of Matter
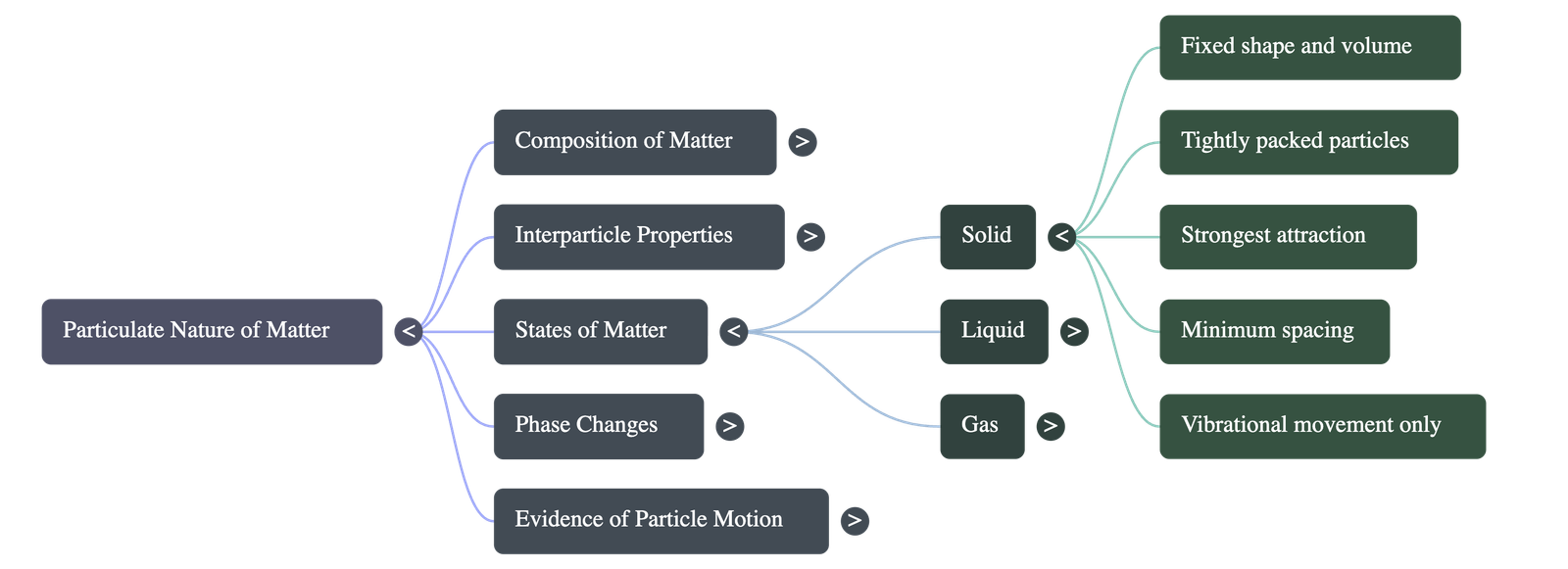
A. Solid State
- Shape and Volume: Solids possess a definite shape and a fixed volume.
- Particle Arrangement: Particles are tightly and closely packed together. Interparticle spaces are at their minimum.
- Forces and Movement: Interparticle attractive forces are maximum. Particles cannot move freely past one another; they can only vibrate or oscillate in their fixed positions.
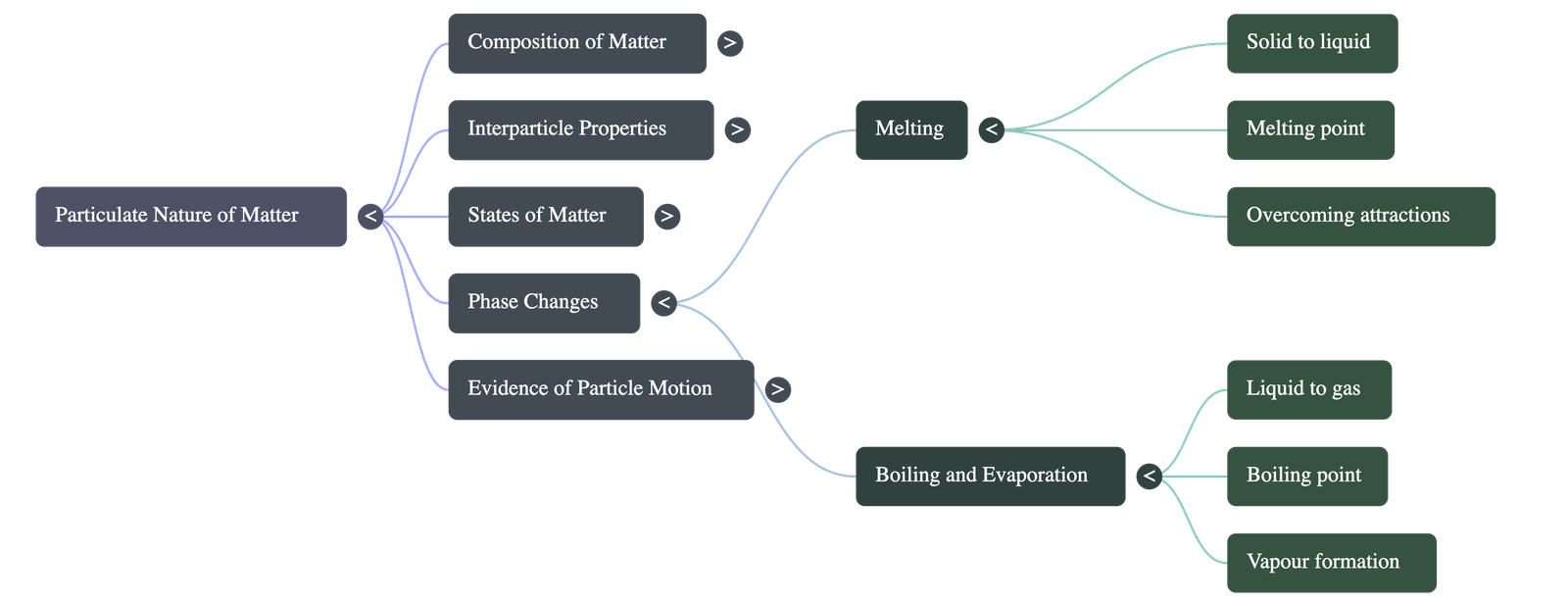
- Melting Point: The minimum temperature at which a solid overcomes these forces and turns into a liquid at atmospheric pressure is called its melting point.
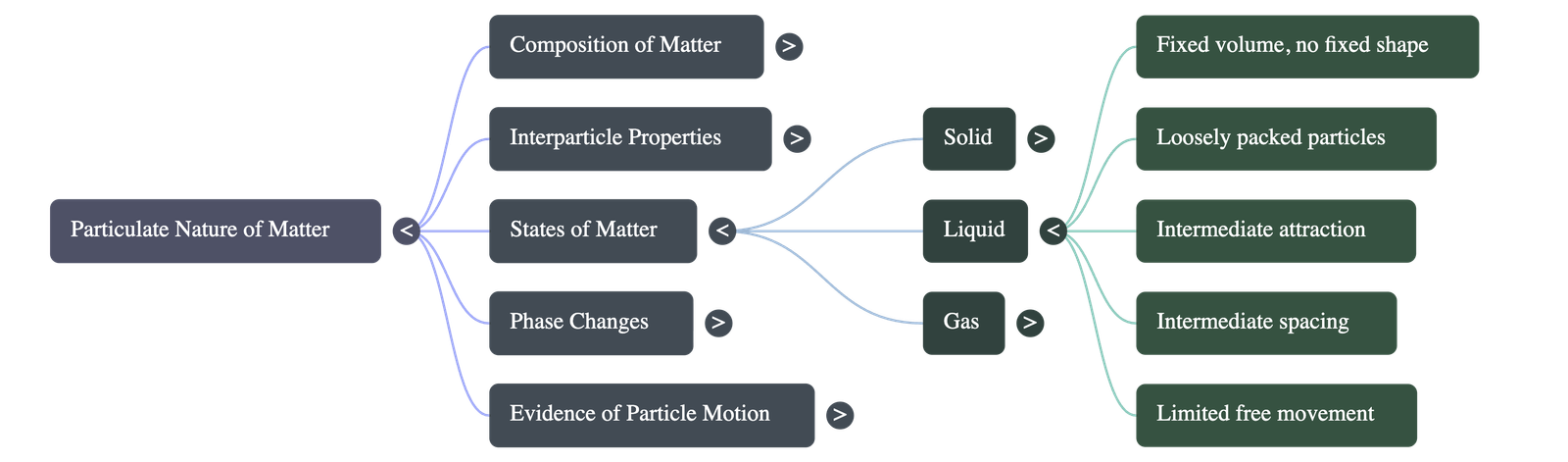
B. Liquid State
- Shape and Volume: Liquids have a definite fixed volume but no fixed shape. They take the shape of the container they are poured into.
- Particle Arrangement: Particles are slightly more loosely packed than in solids, resulting in slightly larger interparticle spaces.
- Forces and Movement: Attractive forces are weaker than in solids but strong enough to keep particles together. Particles can move freely but only within a limited space, allowing the liquid to flow.
- Boiling Point: The temperature at which a liquid turns into a vapour (gas) rapidly at atmospheric pressure is its boiling point.
C. Gaseous State
- Shape and Volume: Gases have neither a fixed shape nor a fixed volume. They will expand to completely fill any container they are enclosed in.
- Particle Arrangement: Interparticle spacing is at its maximum. Gases are highly compressible because of the vast empty space between particles.
- Forces and Movement: Interparticle attractions are negligible. Particles move completely freely and rapidly in all available directions.
4. Movement of Particles and Thermal Energy
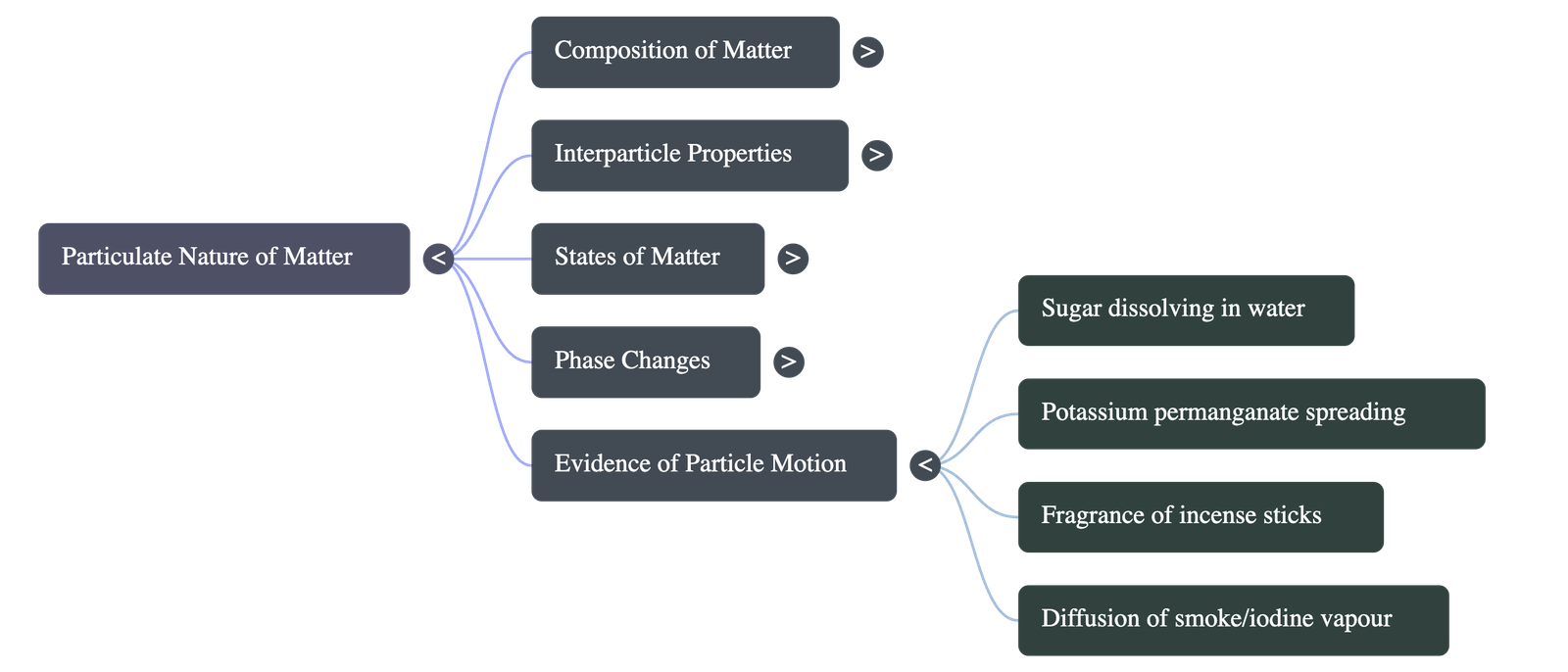
- Constant Motion: Particles of matter are in a state of continuous movement across all states (vibrating in solids, flowing in liquids, darting randomly in gases). For example, the scent of burning incense spreads because constantly moving air particles collide with the fragrance particles and carry them across a room.
- Impact of Heat: The movement of particles drastically increases when heat (thermal energy) is applied. For instance, potassium permanganate crystals dissolve and spread much faster in hot water than in ice-cold water.
- Changing States: The physical state of matter depends on the thermal energy of its particles. Heating a solid gives its particles enough thermal energy to overcome the strong attractive forces, causing it to melt into a liquid. Further heating gives the liquid particles enough energy to completely break free from one another, boiling into a gas.
Quick Navigation:
| | | |
1 / 1
Quick Navigation:
| | | |