Quick Navigation:
| | | |
Electricity: Magnetic and Heating Effects
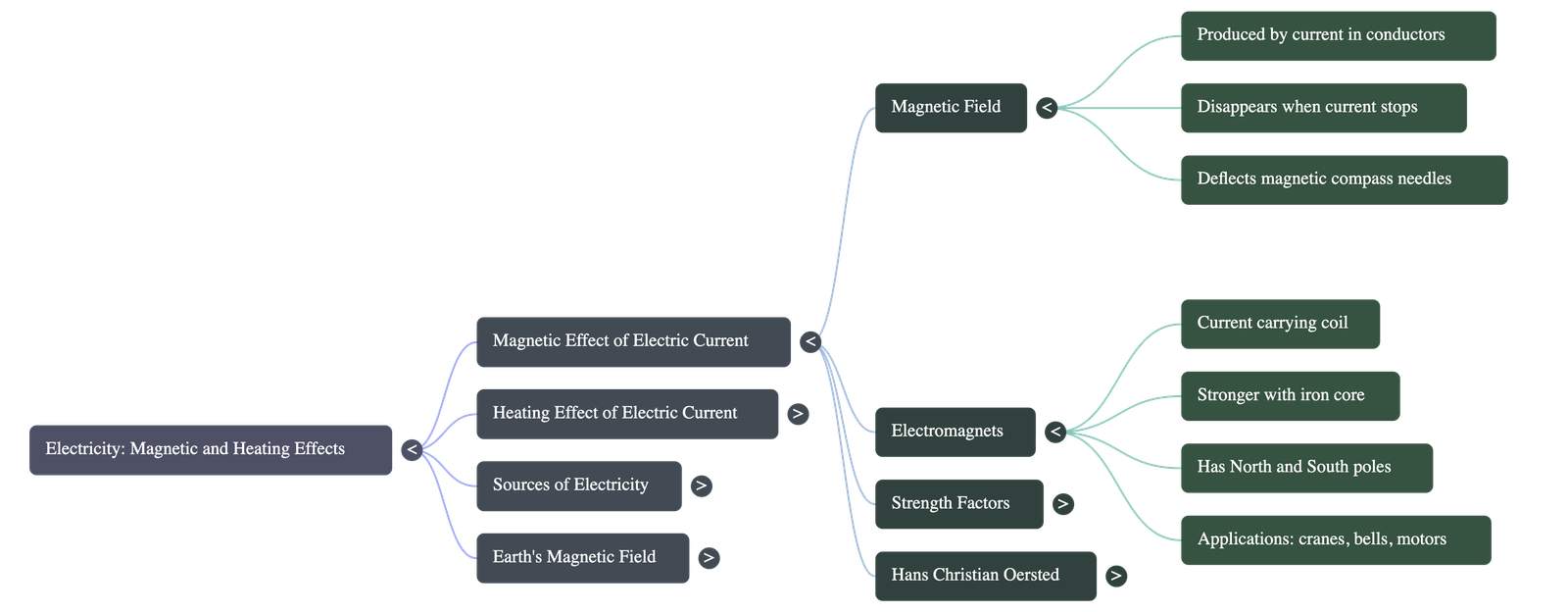
Magnetic Effect of Electric Current
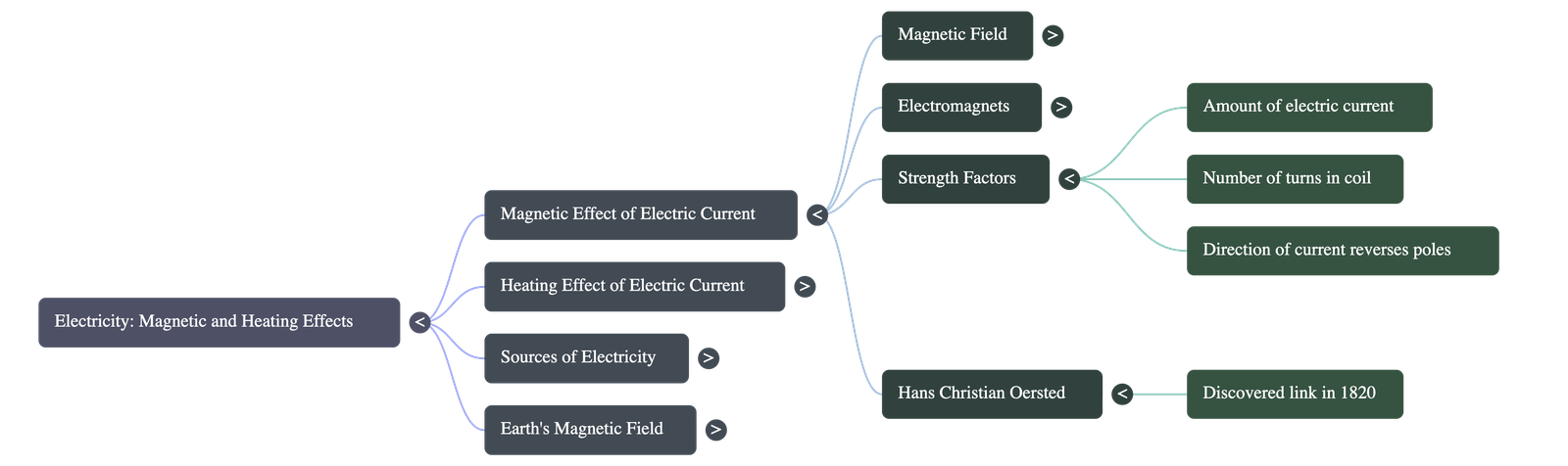
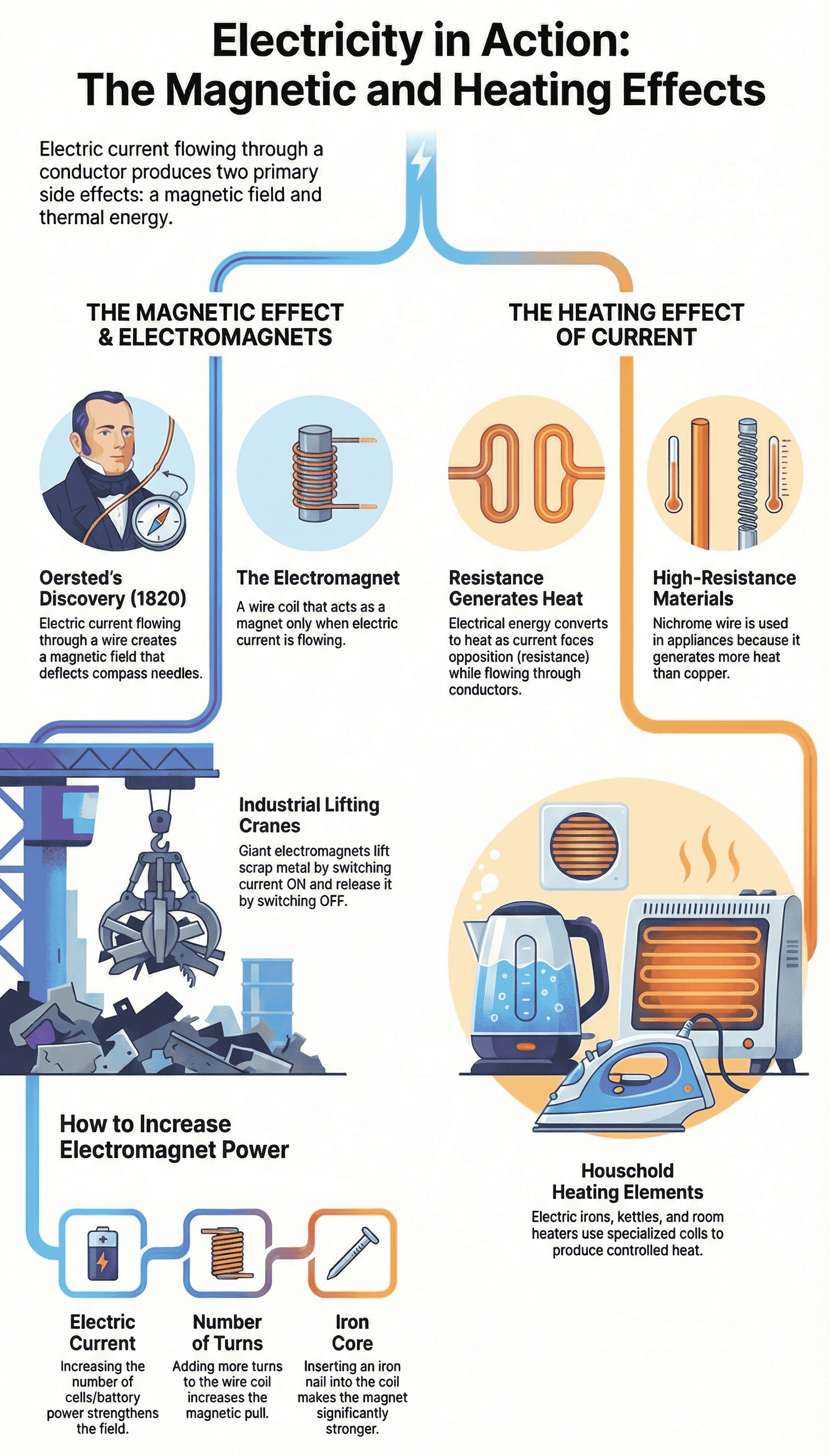
- Discovery: The connection between electricity and magnetism was discovered in 1820 by the Danish scientist Hans Christian Oersted. He noticed that a magnetic compass needle deflected whenever an electrical circuit was opened or closed nearby.
- Basic Principle: When an electric current flows through a conductor, such as a wire, it produces a magnetic field around it. This magnetic effect completely disappears as soon as the electric current stops flowing.
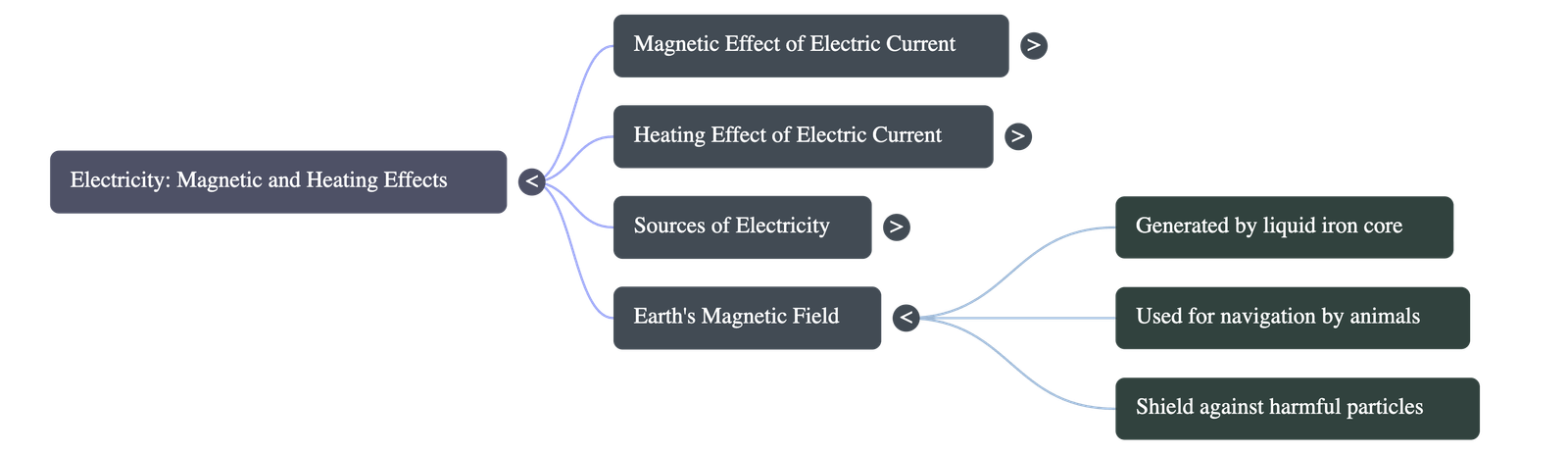
- Earth as a Magnet: The Earth itself behaves like a giant magnet. This is caused by the movement of liquid iron deep inside the Earth's core, which creates electric currents that generate a magnetic field. This field acts as a shield blocking harmful space particles and helps migratory animals and birds navigate.
Electromagnets
- Definition: A current-carrying coil of wire that behaves like a magnet is called an electromagnet.
- Iron Core: Inserting an iron nail or core into the center of the coil makes the electromagnet significantly stronger for practical applications.
- Polarity: Just like permanent magnets, electromagnets have both North and South poles. The poles can be easily reversed by changing the direction of the electric current.
- Strength Modification: The magnetic strength of an electromagnet can be increased by either raising the amount of electric current flowing through it (using more cells in a battery) or by increasing the number of turns in the wire coil.
- Lifting Electromagnets: Large, strong electromagnets are attached to cranes in factories and scrapyards to lift and sort heavy iron and steel objects. The operator can easily release the heavy objects simply by switching off the electric current.
Heating Effect of Electric Current
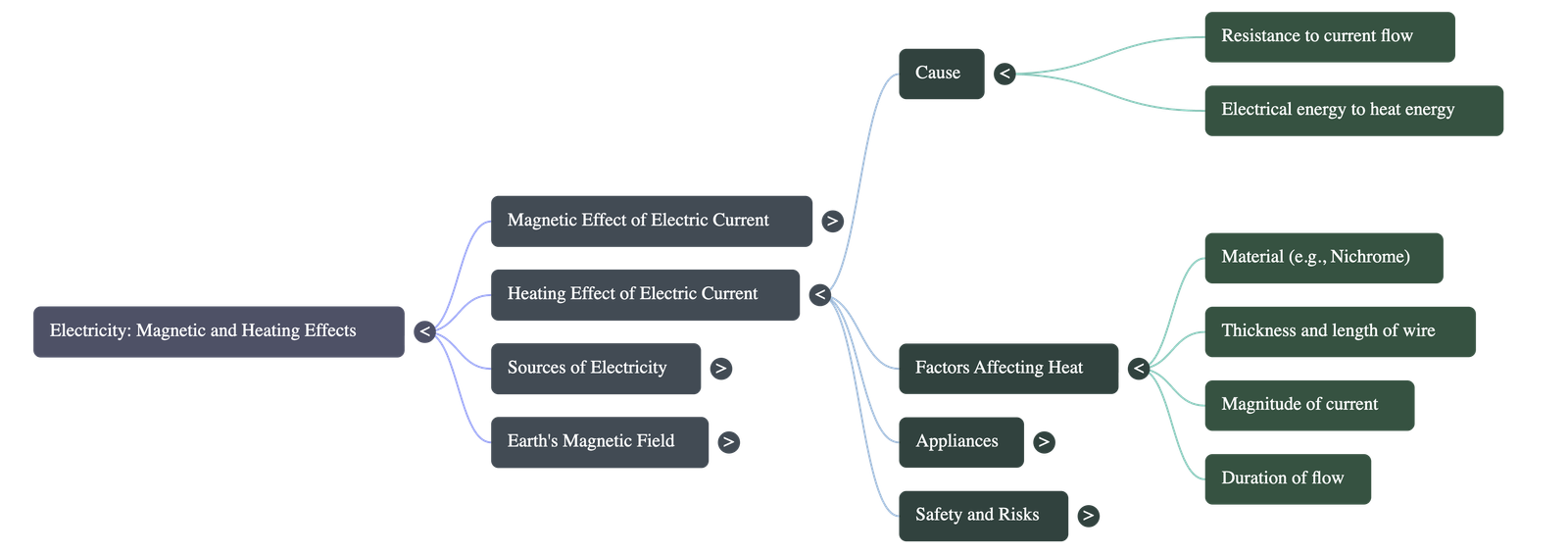
- Basic Principle: When electric current flows through any conductor, it faces resistance. This resistance converts some of the electrical energy into heat energy, causing the conductor to warm up.
- Influencing Factors: The amount of heat generated depends on the material of the wire (for example, nichrome offers higher resistance than copper), its thickness, its length, the magnitude of the electric current, and how long the current flows.
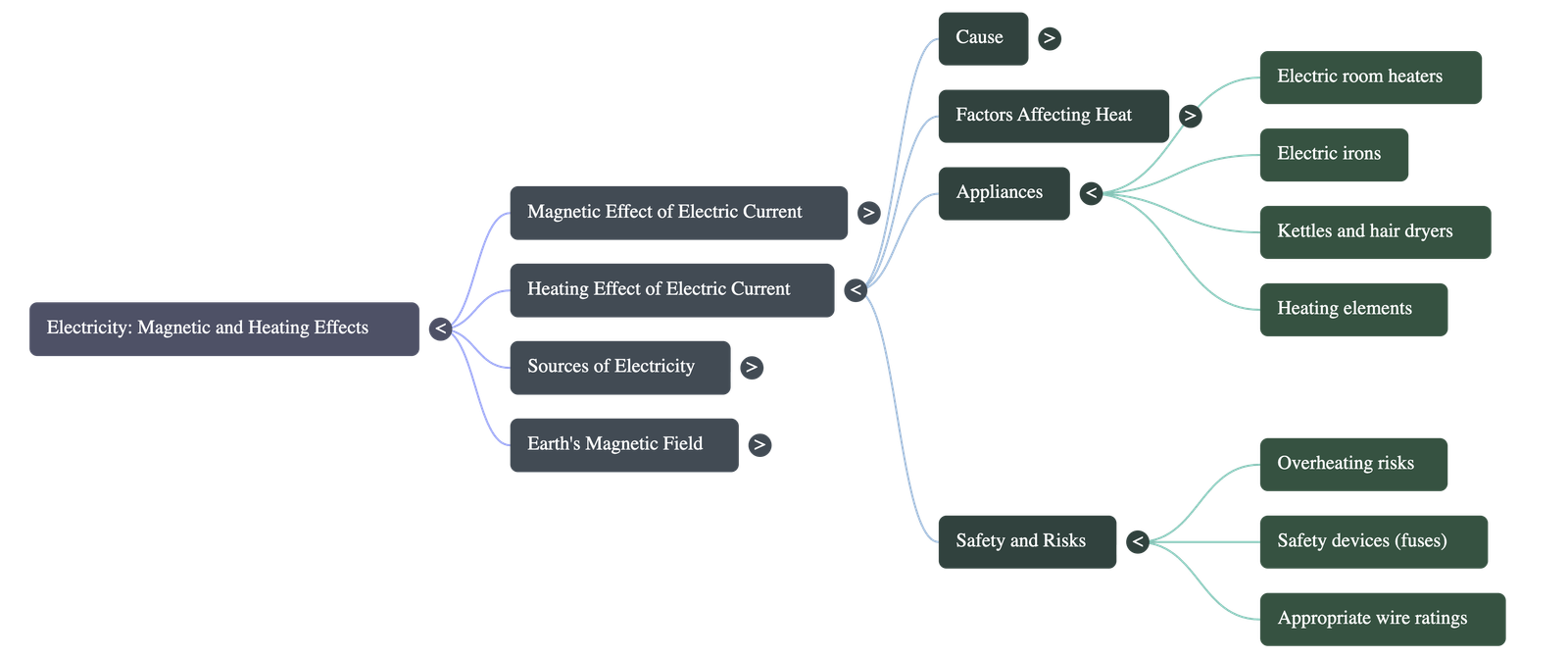
- Everyday Applications: Household appliances like room heaters, electric stoves, kettles, irons, immersion rods, and hair dryers work on this principle. They contain a specific rod or coil called a "heating element" that gets red hot when current passes through it. Incandescent lamps also glow because their filament is heated by an electric current.
- Industrial Use: In the steel manufacturing industry, special high-temperature furnaces use the heating effect of electric current to melt and recycle scrap steel.
- Safety Hazards: Unintended heating can cause energy loss in transmission wires or lead to dangerous situations like melted plastic in plugs and electrical fires. Using properly rated wires, plugs, and sockets is essential for household safety.
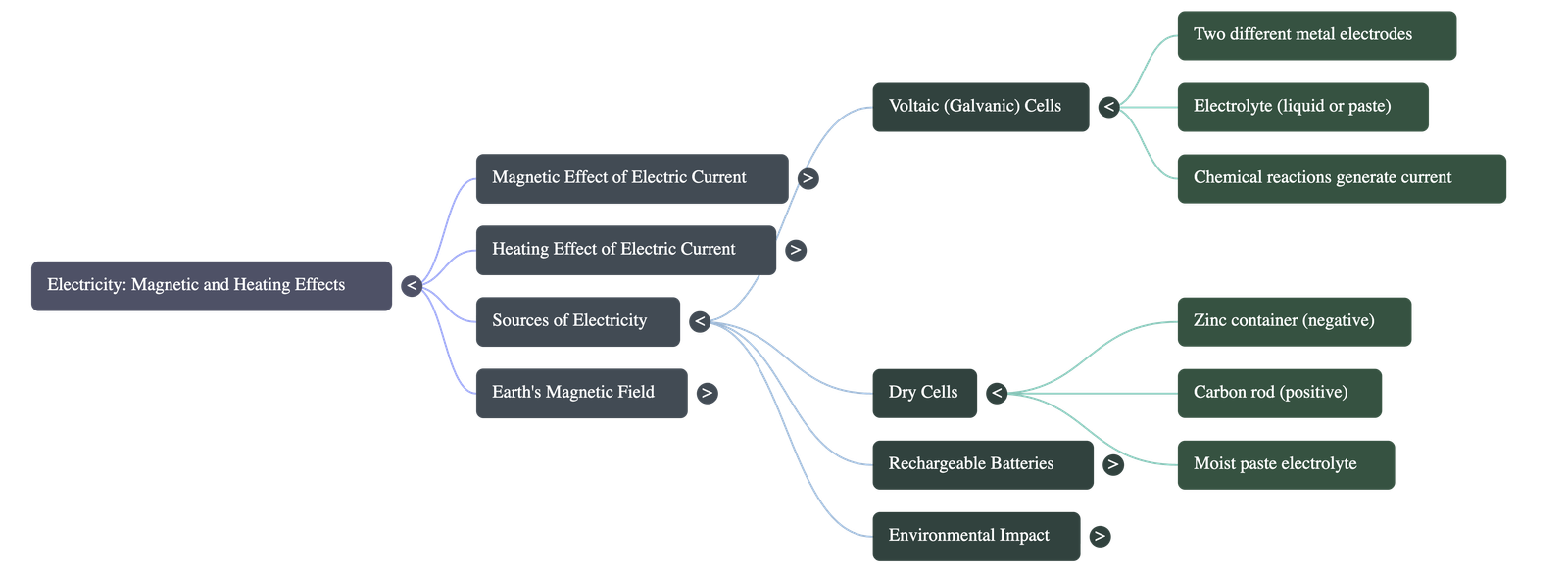
How Batteries Generate Electricity
- Voltaic (Galvanic) Cells: The earliest batteries, developed by Alessandro Volta. They consist of two different metal plates (electrodes) submerged in a liquid called an electrolyte (like a salt solution or weak acid). A chemical reaction between the metals and the liquid produces electricity. A simple DIY fruit battery (like a lemon with copper strips and iron nails) operates on this exact principle.
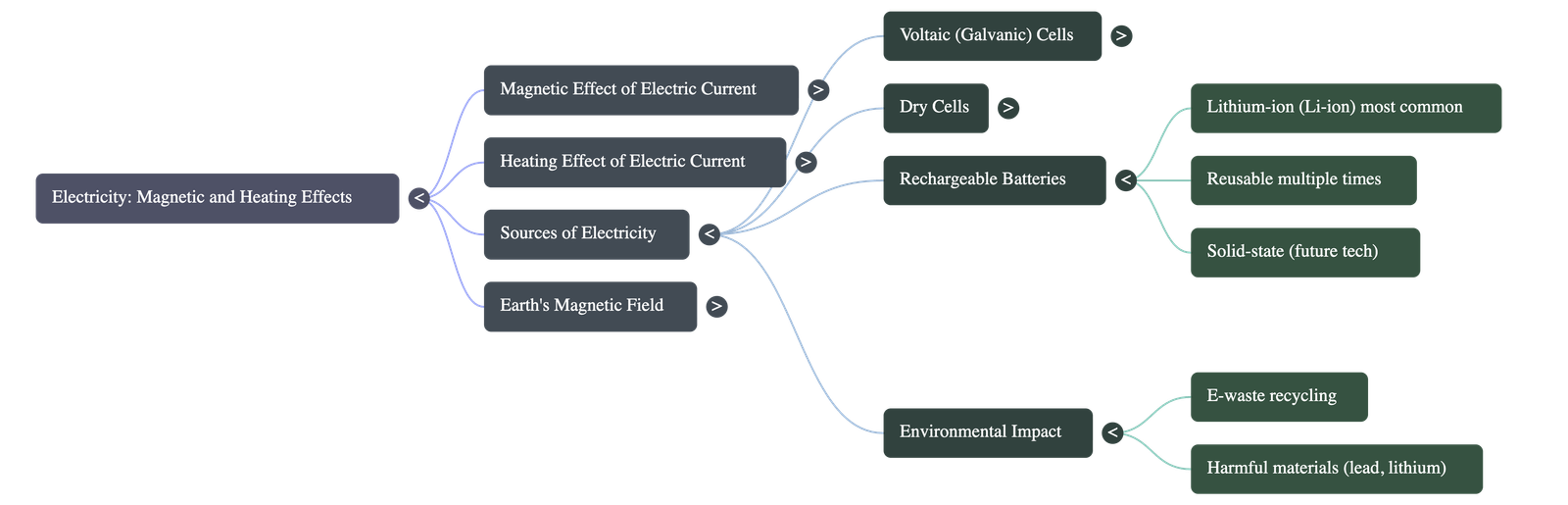
- Dry Cells: Designed for convenience and portability, these cells replace liquid electrolytes with a thick, moist paste. They feature a zinc container acting as the negative terminal and a central carbon rod with a metal cap acting as the positive terminal. They are single-use and die when their chemicals are depleted.
- Rechargeable Batteries: Found in mobile phones, laptops, and electric vehicles, these batteries can be recharged and reused multiple times. Lithium-ion (Li-ion) is currently the most common type. Scientists are also developing next-generation solid-state batteries for faster charging, longer life, and better safety.
- E-Waste and Recycling: Used batteries contain hazardous chemicals (acids) and valuable metals (lithium, cobalt, lead, cadmium, nickel). They should never be thrown in regular garbage but must be taken to specialized e-waste recycling facilities to protect the environment and recover valuable resources.
Quick Navigation:
| | | |
1 / 1
Quick Navigation:
| | | |