Quick Navigation:
| | | |
Nature of Matter: Elements, Compounds, and Mixtures
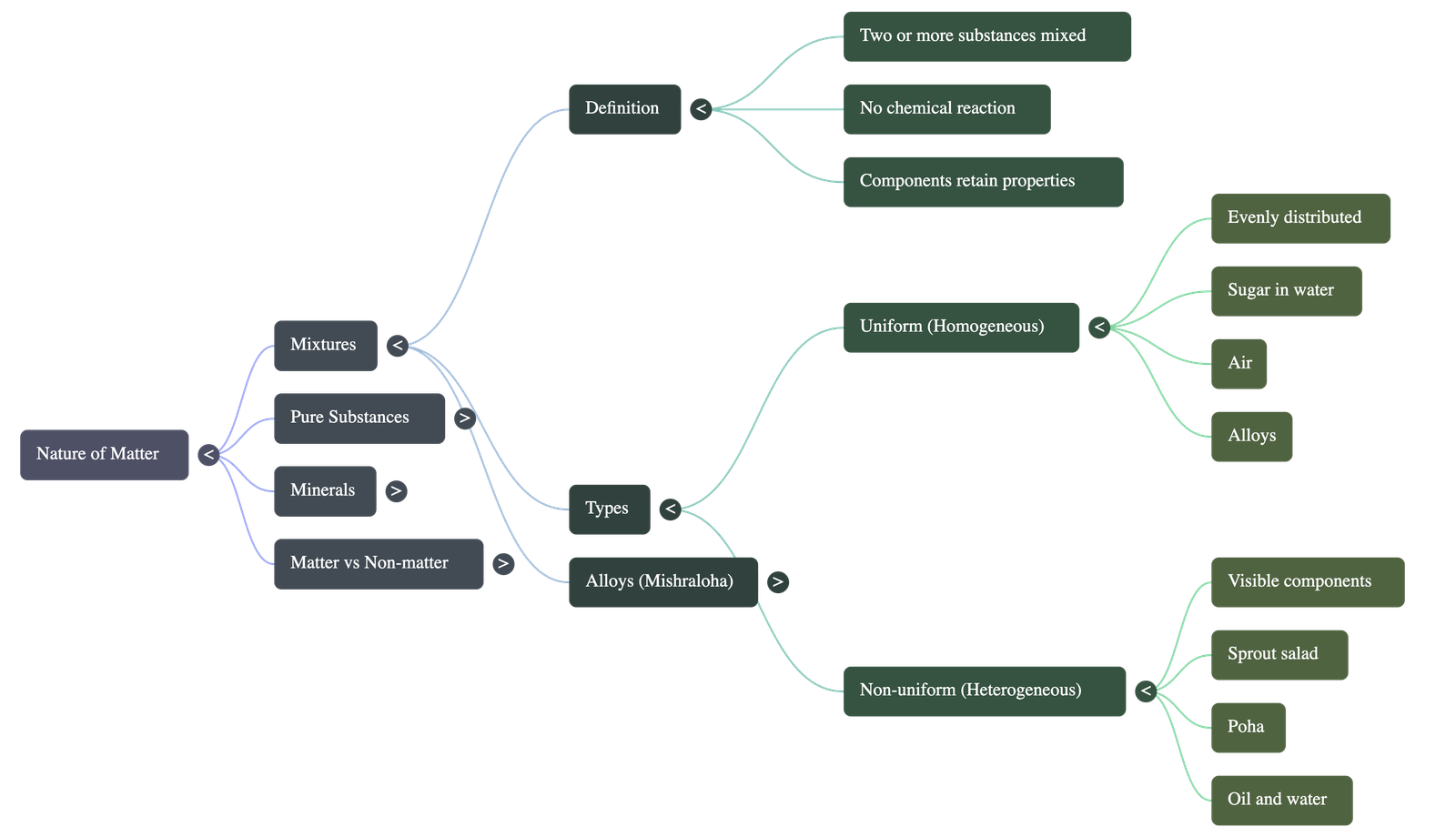
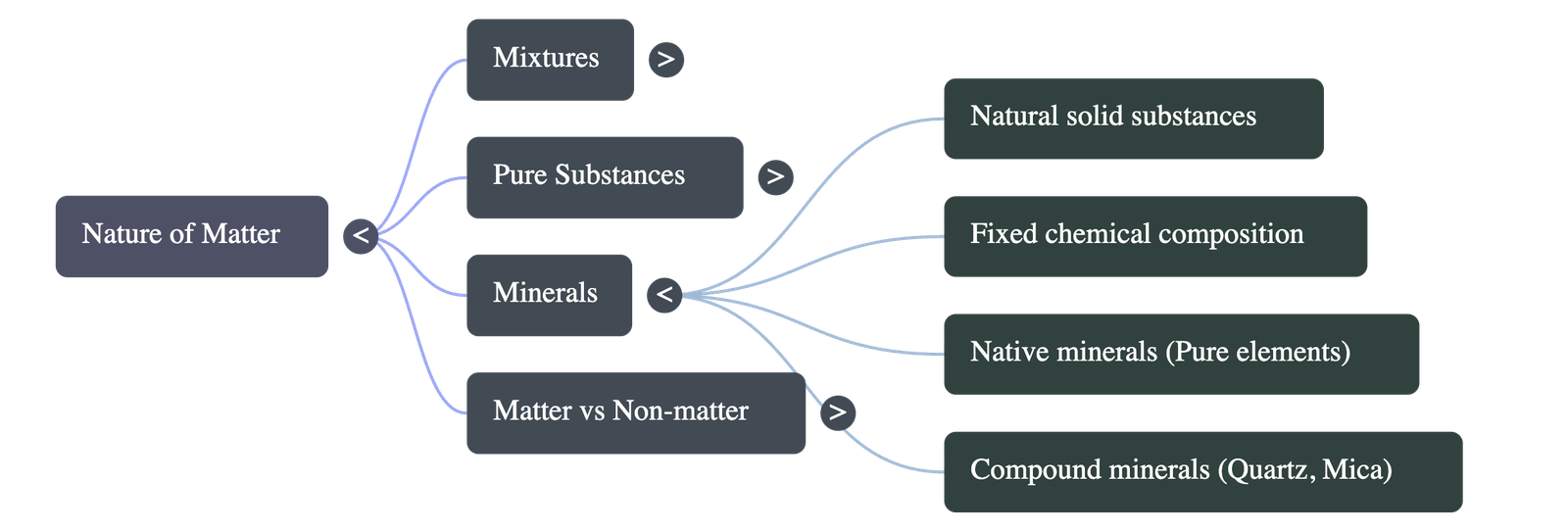
1. What Are Mixtures?
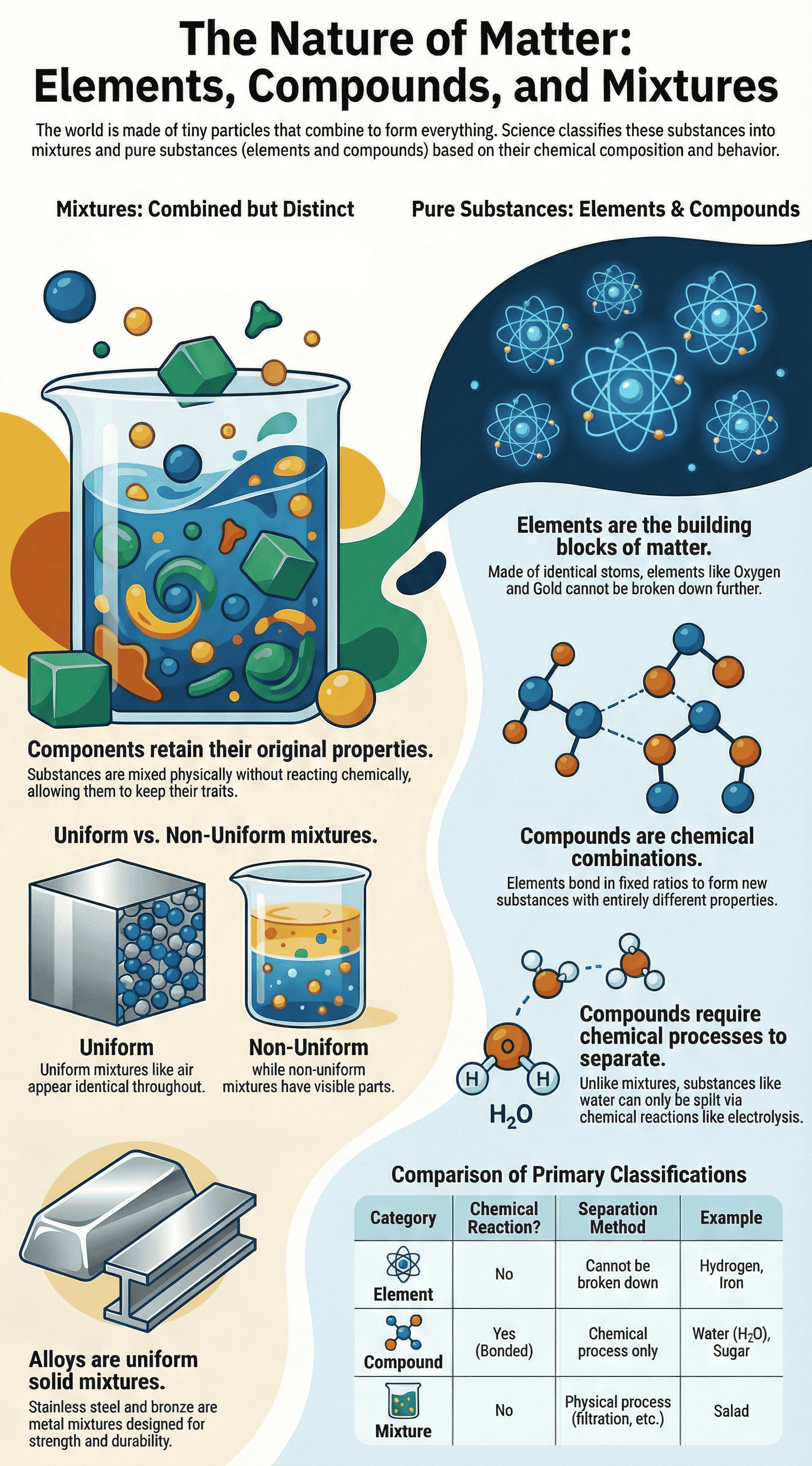
- Definition: A mixture is formed when two or more substances are combined without a chemical reaction. Each substance (component) retains its own individual properties.
- Non-Uniform Mixtures: The different components are not evenly distributed and are generally visible to the naked eye or a magnifying device (e.g., sprout salad, sand and water).
- Uniform Mixtures: The components are evenly distributed and cannot be distinguished even under a microscope (e.g., sugar dissolved in water, air).
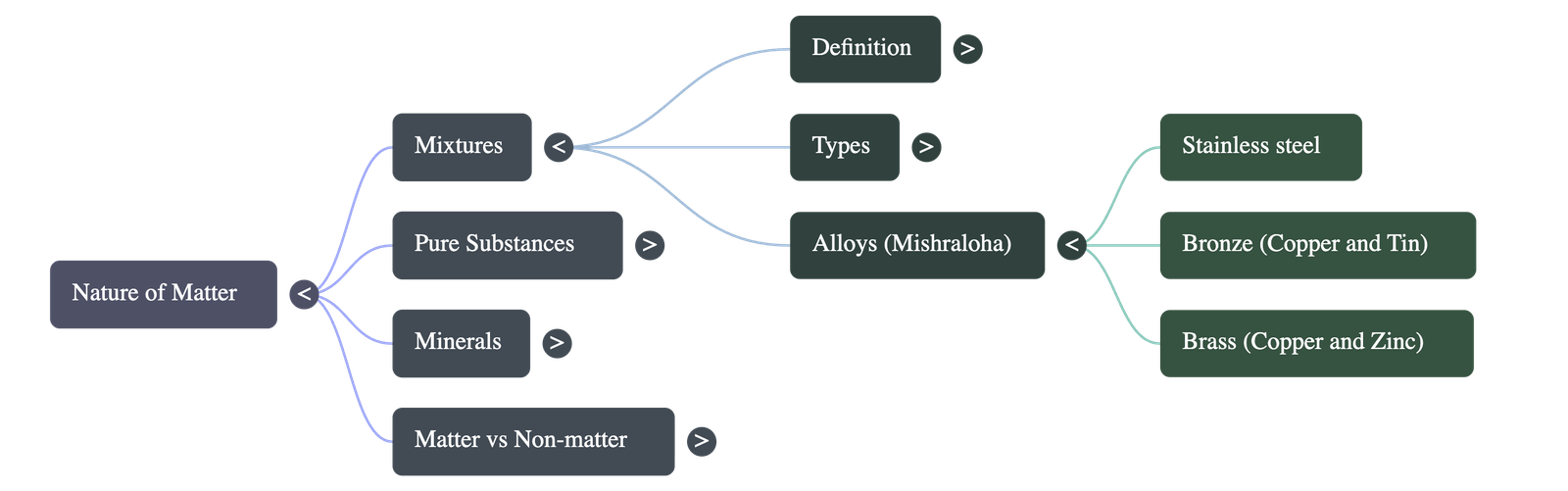
- Alloys: Uniform mixtures of metals (and sometimes non-metals). Known in ancient Indian texts as Mishraloha, examples include Bronze (copper + tin), Brass (copper + zinc), and Stainless Steel (iron + nickel + chromium + carbon).
- Air as a Mixture: Air is a uniform mixture of gases primarily consisting of nitrogen (78%), oxygen, argon, carbon dioxide, and water vapor. Dust particles and gases like carbon monoxide and ozone can also be present as pollutants.
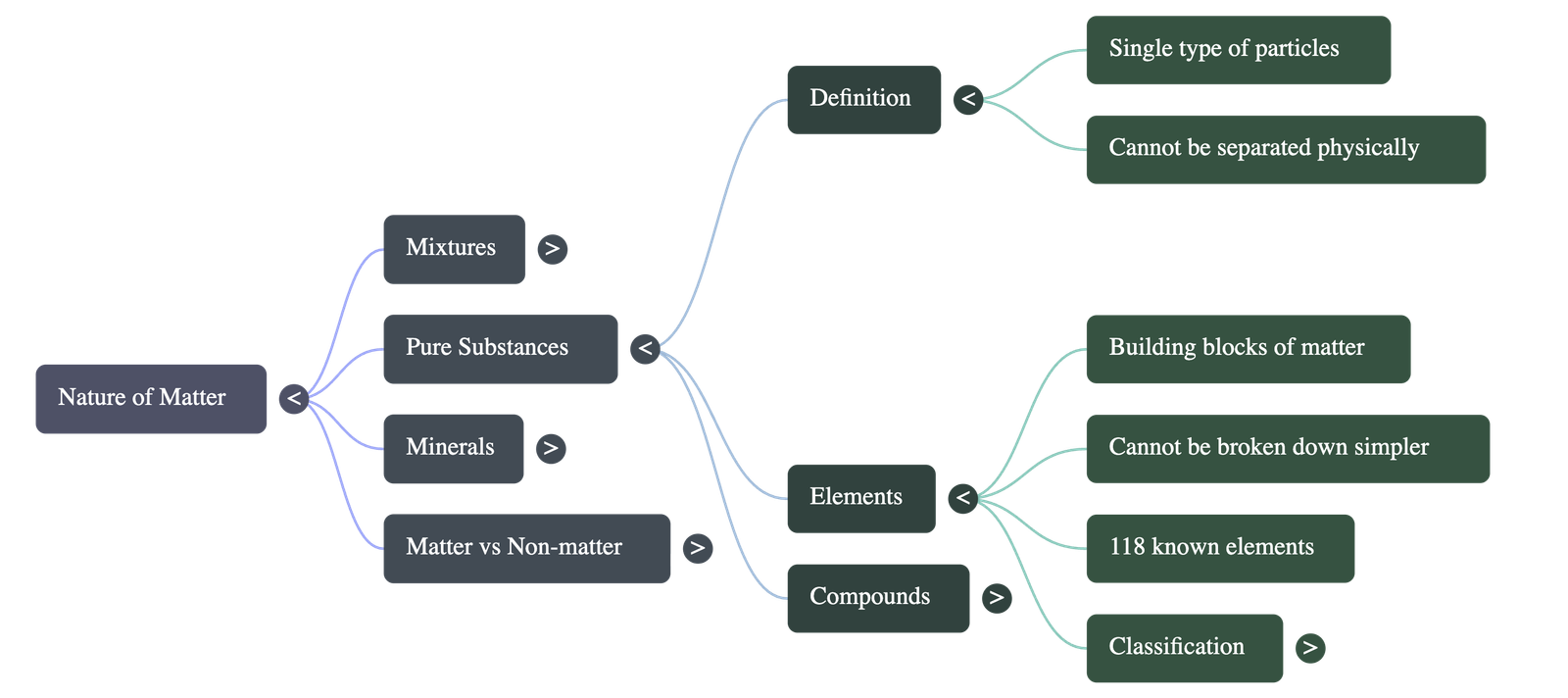
2. What Are Pure Substances?
- Everyday vs. Scientific Meaning: In everyday language, "pure" means unadulterated. In science, a pure substance contains only one kind of matter (identical particles) and cannot be separated into other kinds of matter by physical processes.
- Classification: Pure substances are classified into two main categories: elements and compounds.
3. Elements: The Building Blocks
- Definition: Elements are pure substances that cannot be broken down further into simpler substances. They are made up of identical atoms.
- Molecules: Since most atoms cannot exist independently, two or more atoms of an element often combine to form a stable particle called a molecule (e.g., oxygen gas consists of molecules made of two oxygen atoms).
- Categories: Elements are broadly classified as metals (gold, silver, iron), non-metals (oxygen, sulfur, carbon), and metalloids (silicon, boron).
- States of Matter: Of the 118 known elements, most are solid. Eleven are gases at room temperature, and only two (mercury and bromine) are liquids. Gallium and caesium become liquids just above room temperature (approx. 30 °C).
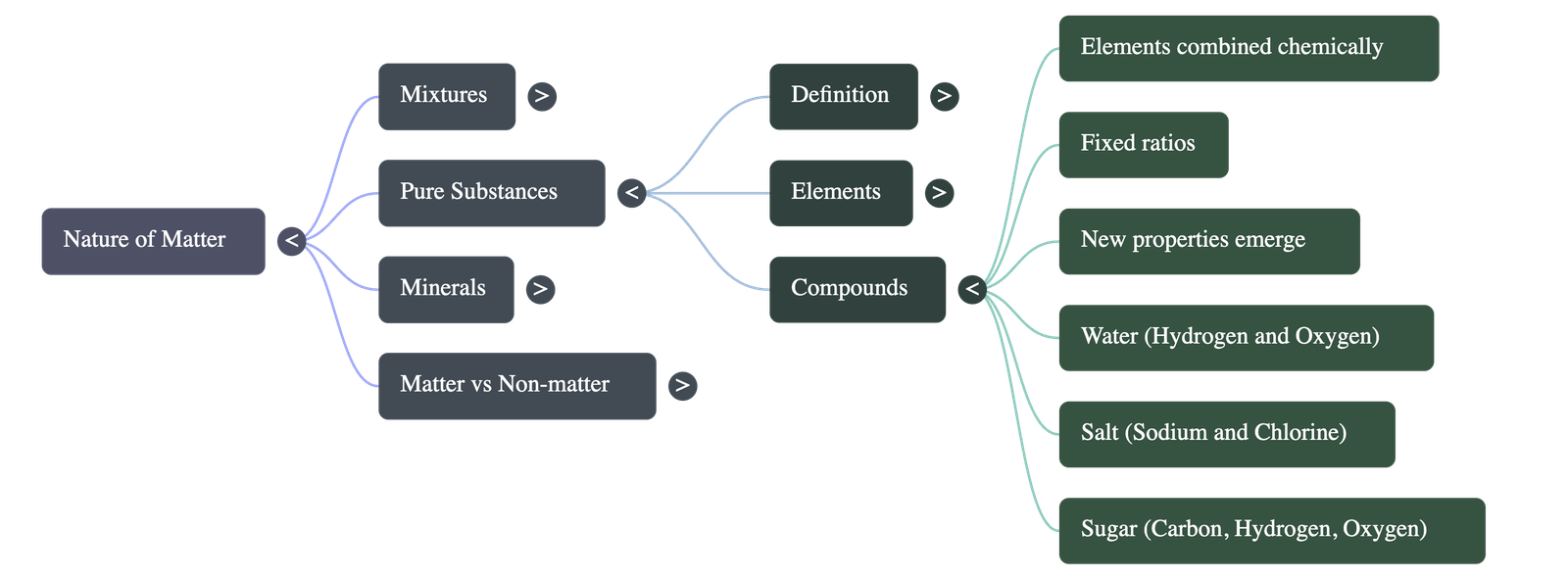
4. Compounds: Chemical Combinations
- Definition: Compounds are formed when different elements chemically combine in fixed, specific ratios to create a completely new substance.
- Distinct Properties: The properties of a compound are entirely different from the elements that make it. For example, water extinguishes fire, even though it is made from hydrogen (a flammable gas) and oxygen (which supports combustion).
- No Physical Separation: Unlike mixtures, the elements in a compound cannot be separated by physical methods. For instance, passing electricity through water is required to separate it into hydrogen and oxygen gases.
- Examples: Common salt (sodium chloride in a 1:1 ratio) and Sugar (decomposes into carbon and water upon heating).
5. Mixtures vs. Compounds: An Experiment
Heating iron filings and sulfur powder reveals the difference between a mixture and a compound:
- As a Mixture (Before Heating): The iron and sulfur retain their properties. Iron can be physically pulled out using a magnet. Adding hydrochloric acid produces an odorless hydrogen gas (which burns with a pop sound).
- As a Compound (After Heating): A black mass called iron sulfide is formed. It has entirely new properties, is completely unaffected by a magnet, and reacts with hydrochloric acid to release hydrogen sulfide gas (which smells like rotten eggs).
6. Applications in the Real World
- Medicine and Agriculture: Chemists apply knowledge of how elements form compounds to create life-saving medicines, vaccines, and crop-enhancing fertilizers.
- Material Science: Understanding mixtures and compounds allows engineers to create durable alloys like stainless steel and innovative materials like graphene aerogel (the lightest material on Earth, excellent for cleaning oil spills).
- Cultural Heritage: The Indian Dhokra art leverages alloys like brass and bronze, cast into beautiful figures utilizing traditional wax-molding techniques.
7. Minerals
- Definition: Minerals are solid, naturally occurring substances on Earth with fixed chemical compositions.
- Mineral Compounds: Most minerals are compounds made of more than one element (e.g., quartz, calcite, mica, talc, which is used to make talcum powder).
- Native Minerals: Some minerals occur as pure elements, meaning they are not compounds. Examples include pure metals like gold, silver, and copper, and non-metals like sulfur and carbon.
Quick Navigation:
| | | |
1 / 1
Quick Navigation:
| | | |