Quick Navigation:
| | | |
Heat and Energy
1. Heat and Temperature
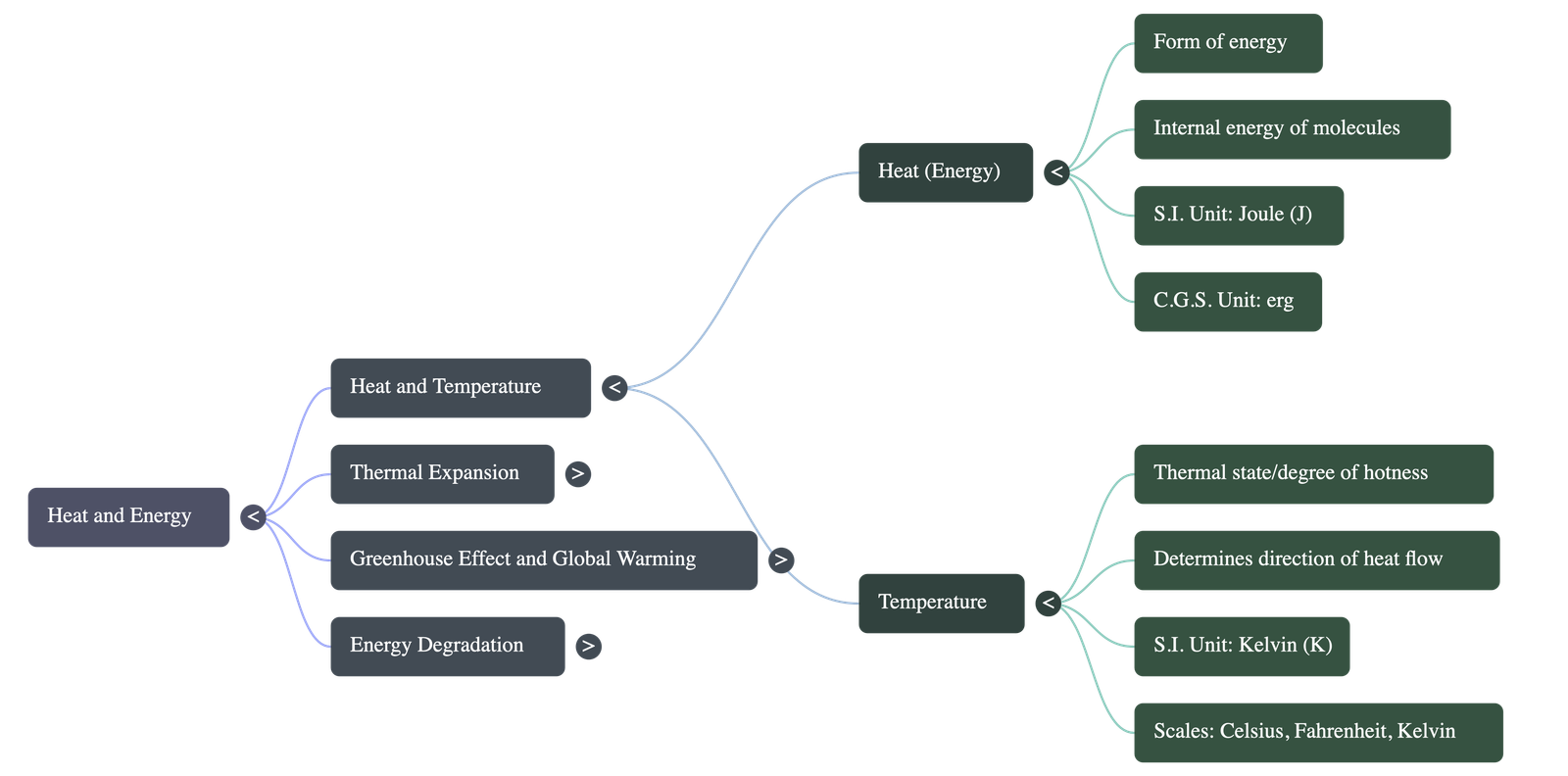
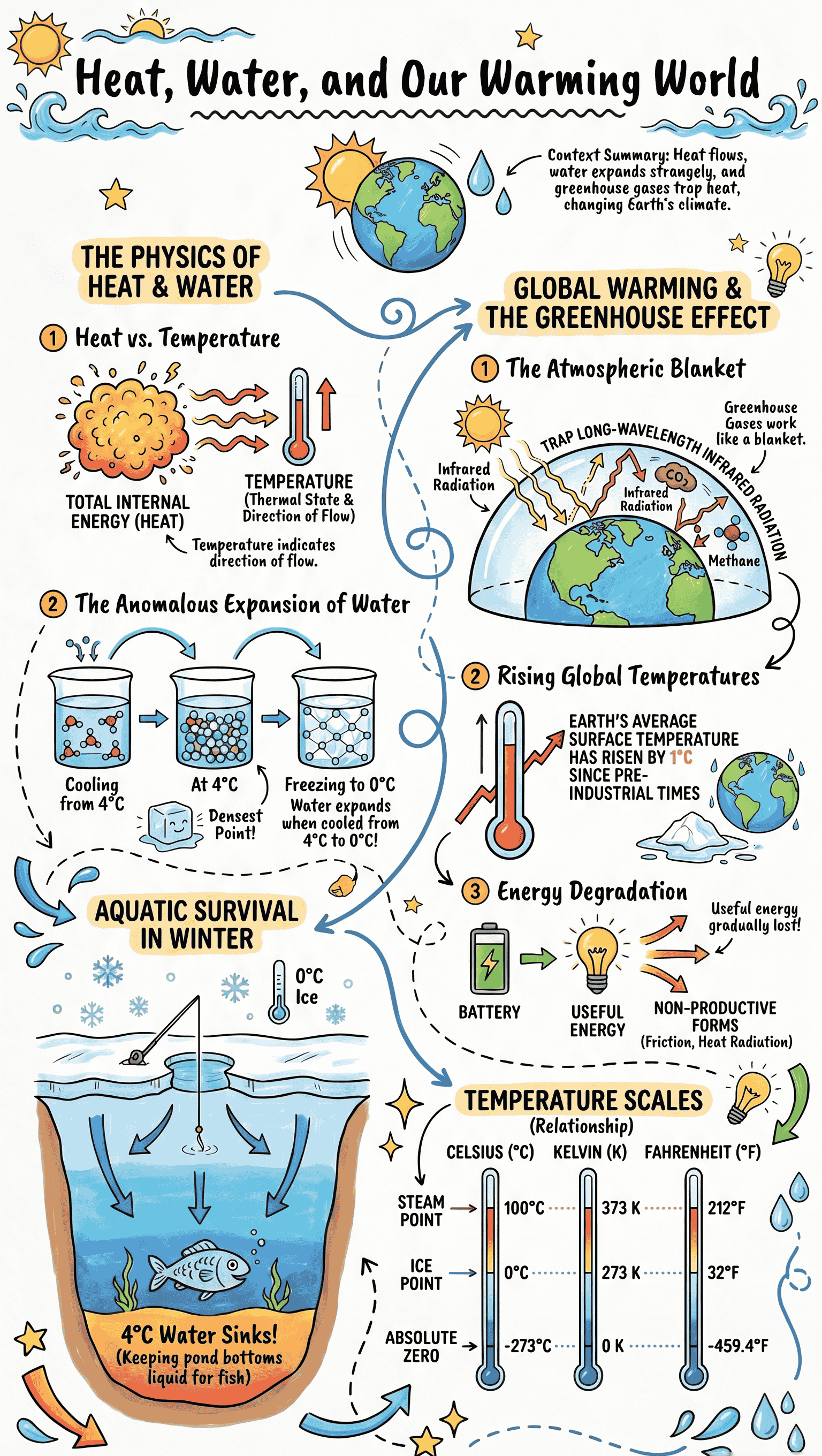
- Concept of Heat: Heat is the form of energy that is transferred between two bodies at different temperatures when placed in contact. It arises from the random motion of molecules within a substance.
- Units of Heat: The S.I. unit of heat is the joule. Other common units include the calorie and kilocalorie.
- Concept of Temperature: Temperature indicates the thermal state (degree of hotness or coldness) of a body. It dictates the direction of heat flow, which always moves from a body at a higher temperature to one at a lower temperature.
- Units of Temperature: The S.I. unit is the kelvin. Other scales include degrees Celsius and degrees Fahrenheit.
2. Thermal Expansion and Anomalous Expansion of Water
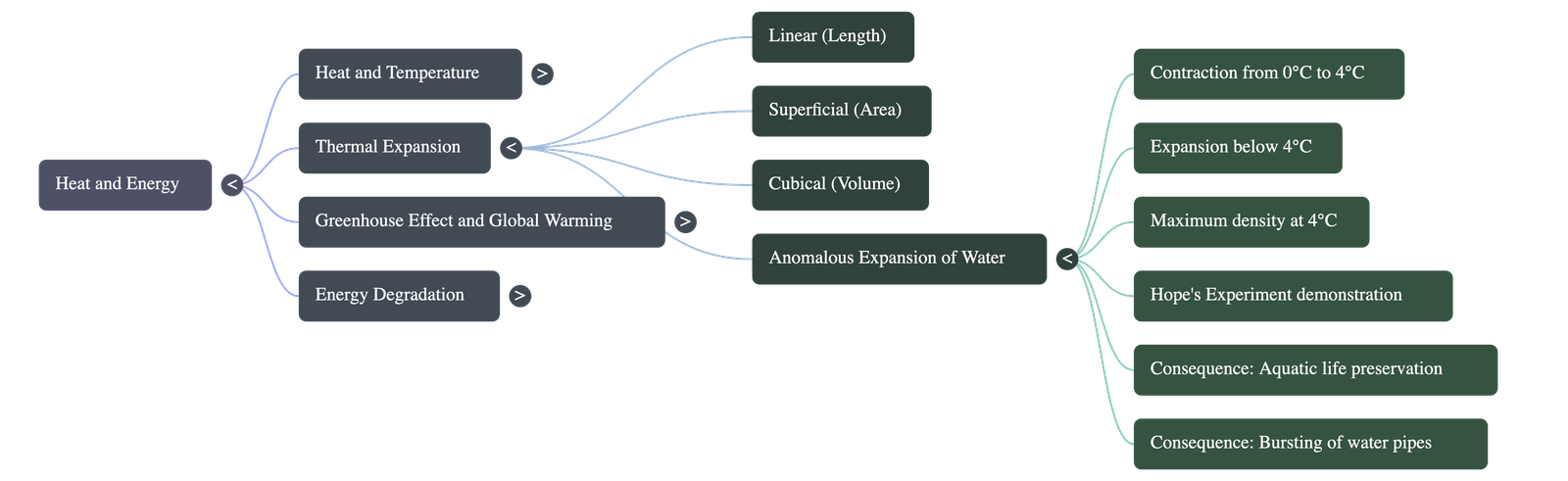
- Thermal Expansion: Most substances expand on heating and contract on cooling. Solids experience linear, superficial, and cubical expansion, whereas liquids and gases only experience cubical expansion.
- Anomalous Expansion: Water behaves unusually when heated from 0 degrees Celsius to 4 degrees Celsius; it contracts instead of expanding. Upon further heating above 4 degrees Celsius, it expands normally.
- Density of Water: Because of its anomalous expansion, water has its minimum volume and maximum density at exactly 4 degrees Celsius.
- Hope's Experiment: Scientist T.C. Hope demonstrated this anomalous behavior using a metallic cylinder fitted with two thermometers and a central cooling trough. The experiment proves that water reaches its maximum density at 4 degrees Celsius and sinks to the bottom.
- Consequences in Nature: The anomalous expansion of water allows aquatic life to survive in frozen ponds during winter, as water at 4 degrees Celsius remains at the bottom while ice forms on the surface. Conversely, it is also responsible for the bursting of water pipelines and destruction of plant crops during very cold nights.
3. Greenhouse Effect
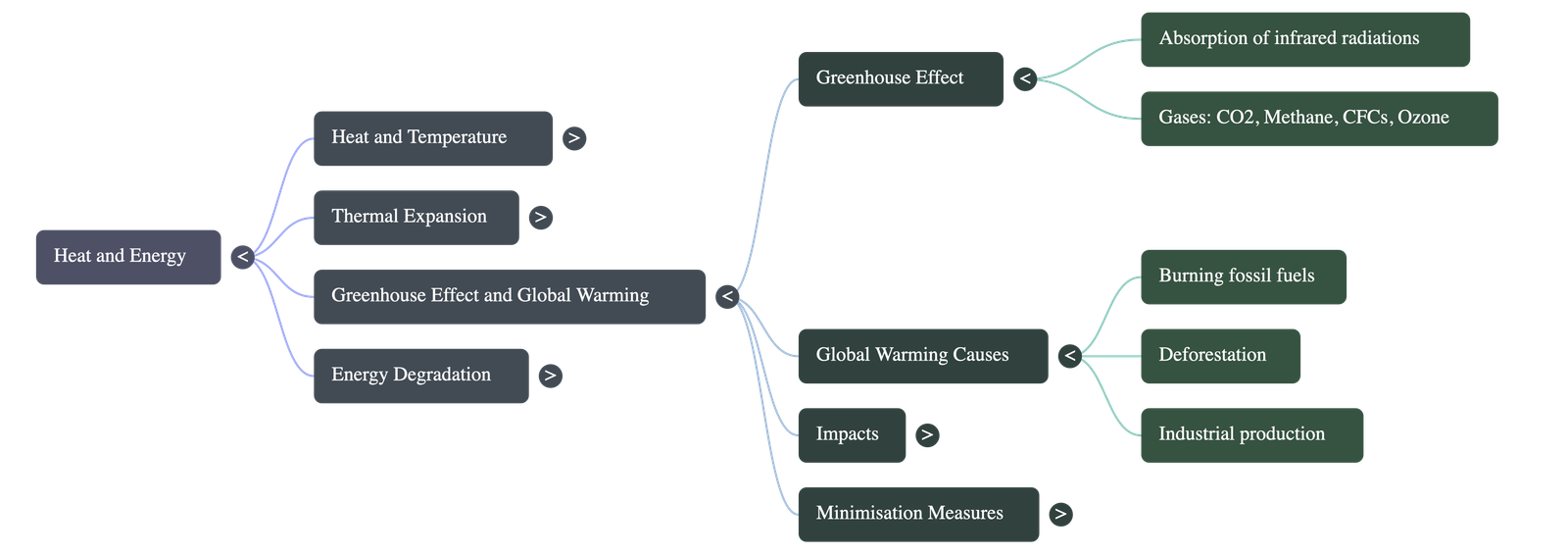
- Definition: The greenhouse effect is the warming of the earth's surface and atmosphere caused by the absorption of long-wavelength infrared radiations emitted from the earth.
- Greenhouse Gases: Gases such as carbon dioxide, methane, nitrous oxide, ozone, and chlorofluorocarbons (CFCs) are excellent absorbers of infrared radiations and trap heat within the atmosphere.
- Natural Necessity: A natural greenhouse effect is essential; without it, the earth's average temperature would drop drastically, making it difficult to sustain a comfortable environment.
4. Global Warming and Its Impacts
- Global Warming: This refers to the abnormal increase in the average effective temperature near the earth's surface due to elevated levels of greenhouse gases resulting from human activities.
- Causes: The primary drivers are the burning of fossil fuels, rapid industrialization, extensive deforestation, and population explosion.
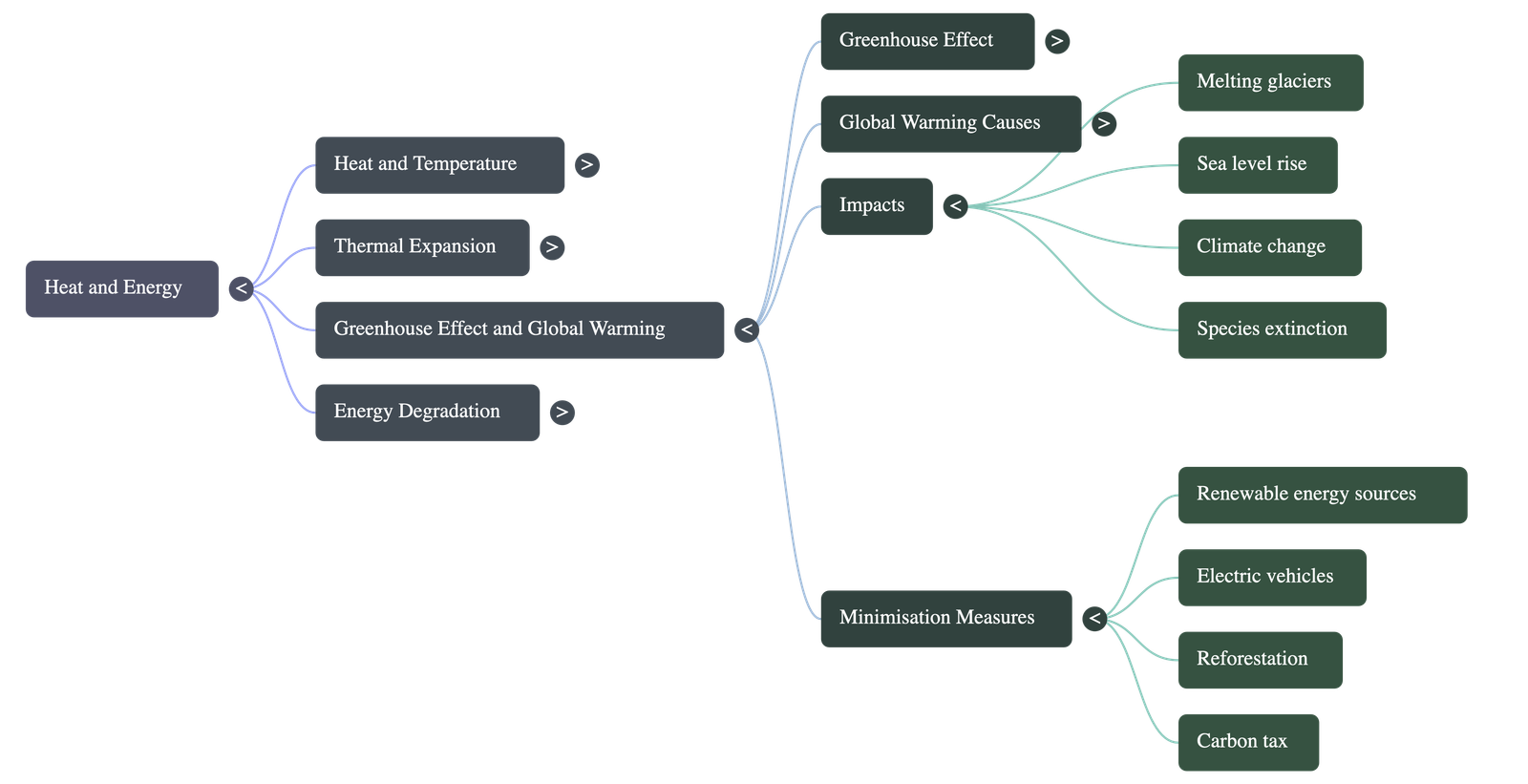
- Major Impacts: Global warming leads to the melting of polar ice caps and glaciers, significant rises in sea levels, warming and acidification of oceans, shifts in farming regions, regional climate disruptions, and the increased spread of heat-related diseases.
5. Measures to Minimise Global Warming
- Technological Measures: Transitioning to renewable sources of energy (solar, wind, etc.) rather than fossil fuels, utilizing battery-operated vehicles, and promoting bio-char stoves for cooking in developing nations.
- Economic Measures: Enforcing widespread reforestation, adopting sustainable land usage practices, and implementing a carbon tax on heavily polluting industries.
- Policy Measures: Educating populations to adopt sustainable lifestyles, encouraging family planning to control population growth, and pushing for global carbon emission reduction agreements.
6. Energy Degradation
- Concept: According to the law of conservation of energy, energy can neither be created nor destroyed. However, during real-world energy transformations, not all input energy converts into useful work.
- Degradation: The gradual decrease of useful energy due to unavoidable factors like friction, radiation loss, and heat dissipation is called the degradation of energy.
- Examples: When lighting a bulb, a massive portion of electrical energy is lost as heat instead of light. Similarly, in vehicles, much of the fuel's energy is wasted as heat combating engine friction rather than contributing to motion.
Quick Navigation:
| | | |
1 / 1
Quick Navigation:
| | | |