Quick Navigation:
| | | |
Study of Gas Laws
1. Introduction to Gases
- State of Matter: Gases lack a definite shape or volume, occupying all the available space within their container.
- Intermolecular Distance: The space between gas molecules is significantly larger compared to liquids, and almost completely negligible in solids.
- Force of Attraction: The attractive forces between gas molecules are negligible.
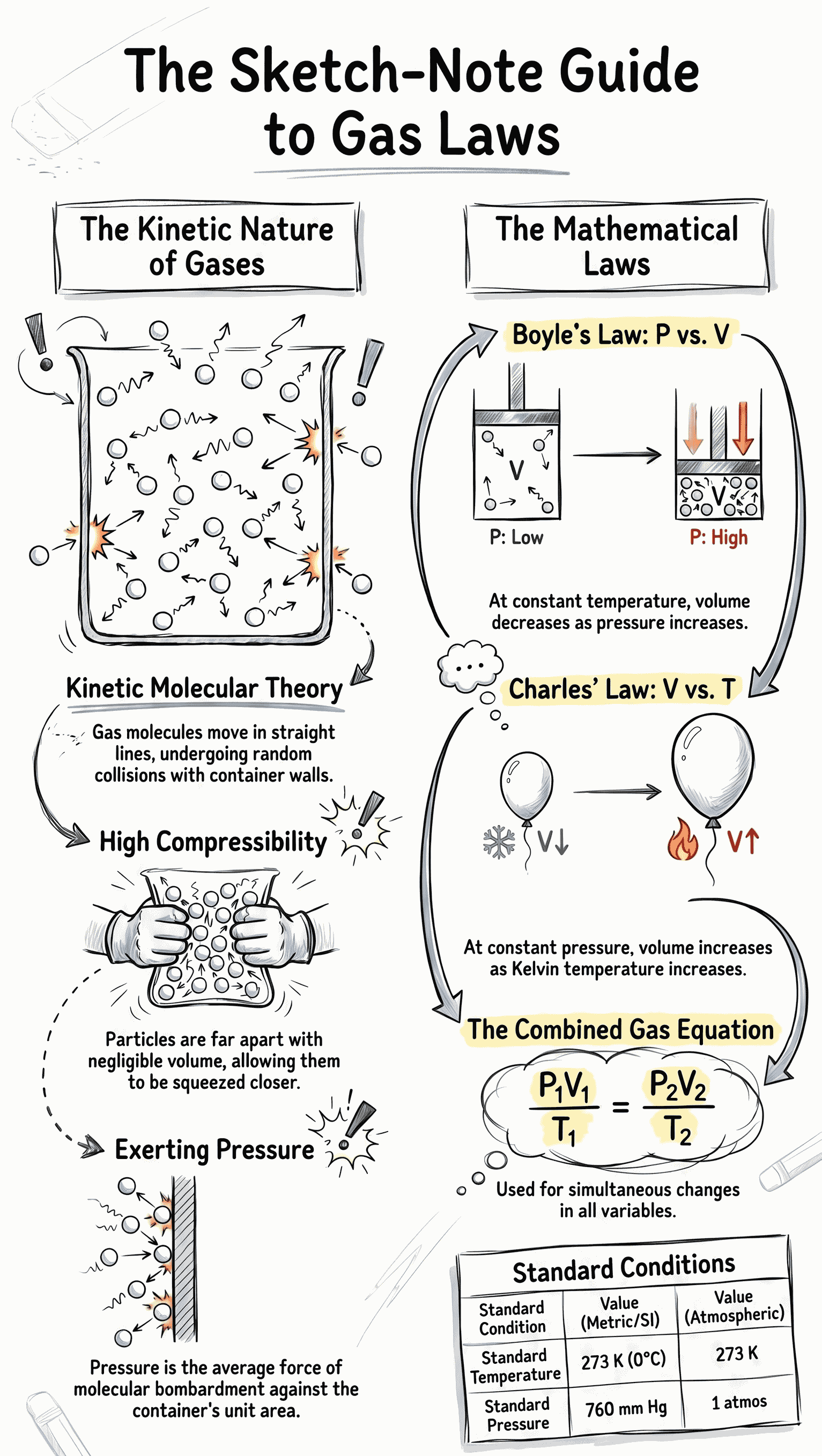
- Molecular Motion: Gas molecules move continuously in straight lines, undergoing random collisions with each other and the container walls.
2. Behaviour & Characteristic Properties
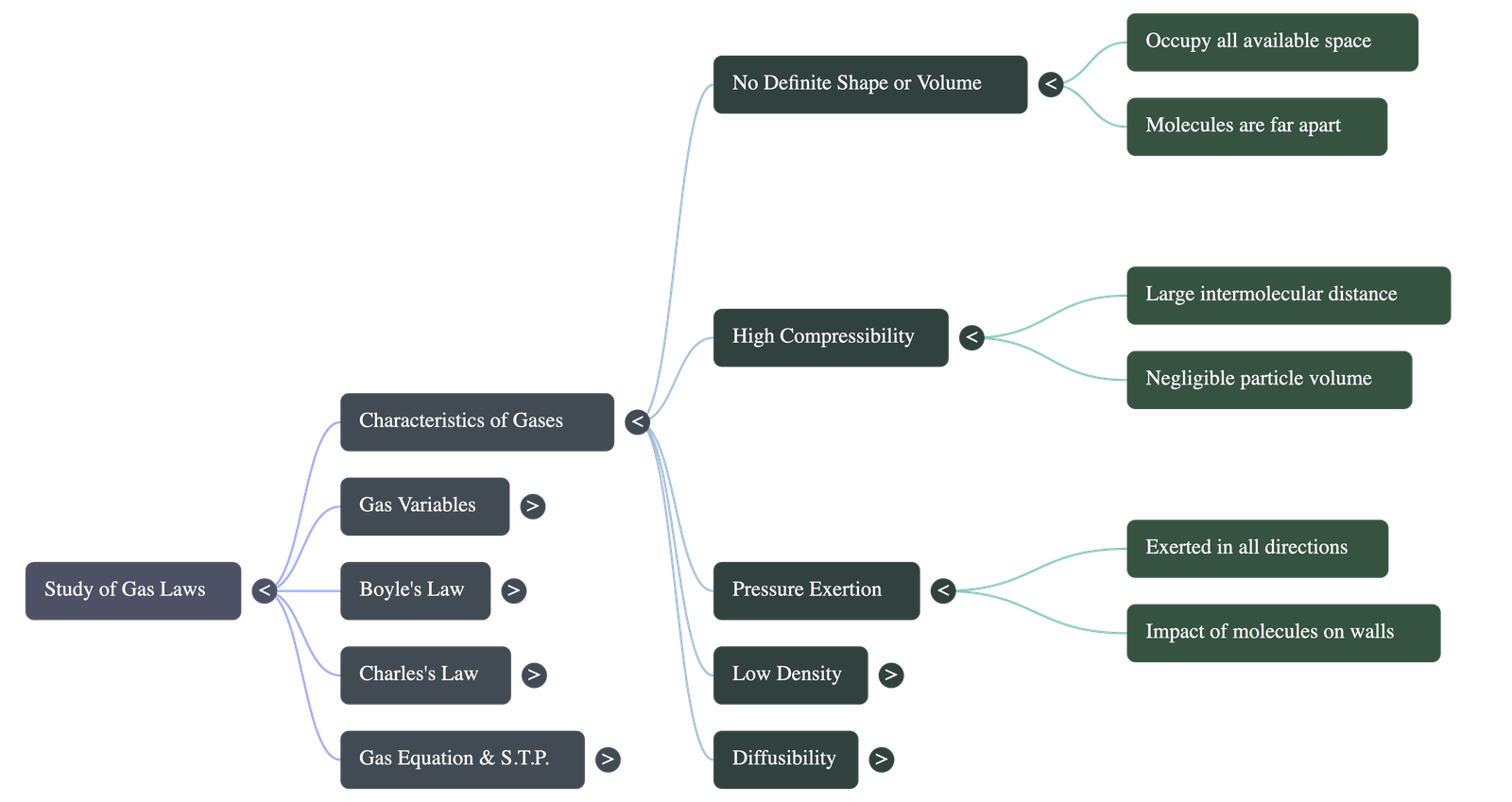
- No Definite Shape or Volume: Due to extremely weak intermolecular forces, molecules move freely and fill the container entirely.
- High Compressibility: Because the actual volume of individual particles is negligible compared to the large intermolecular distances, gases can be easily compressed into much smaller volumes.
- Uniform Pressure: Molecules striking the container walls with high velocity exert an equal and uniform pressure in all directions.
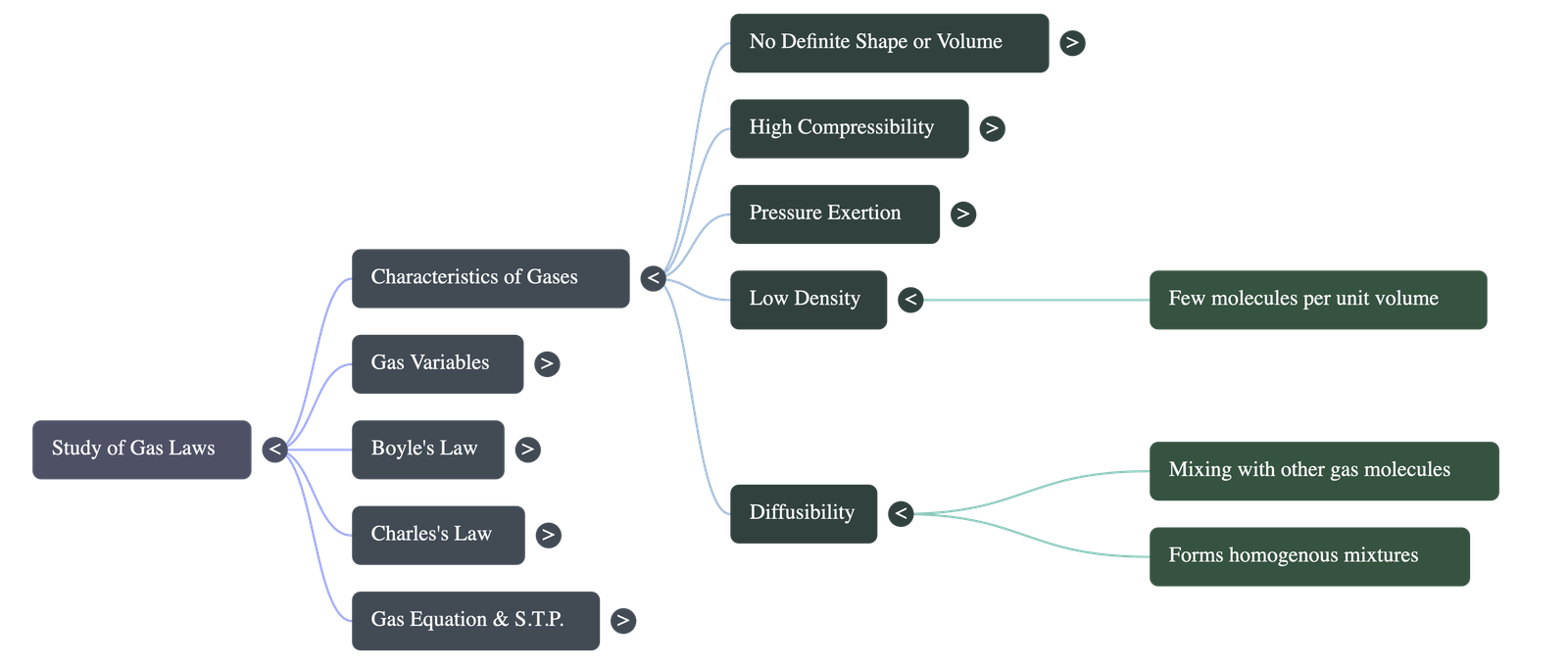
- Low Densities: The massive spaces between molecules mean there are very few molecules per unit volume compared to solids and liquids.
- Diffusibility & Miscibility: Gases readily mix and diffuse into one another to form homogeneous mixtures entirely on their own because of their large intermolecular spaces.
3. Three Variables Governing Gas Behaviour
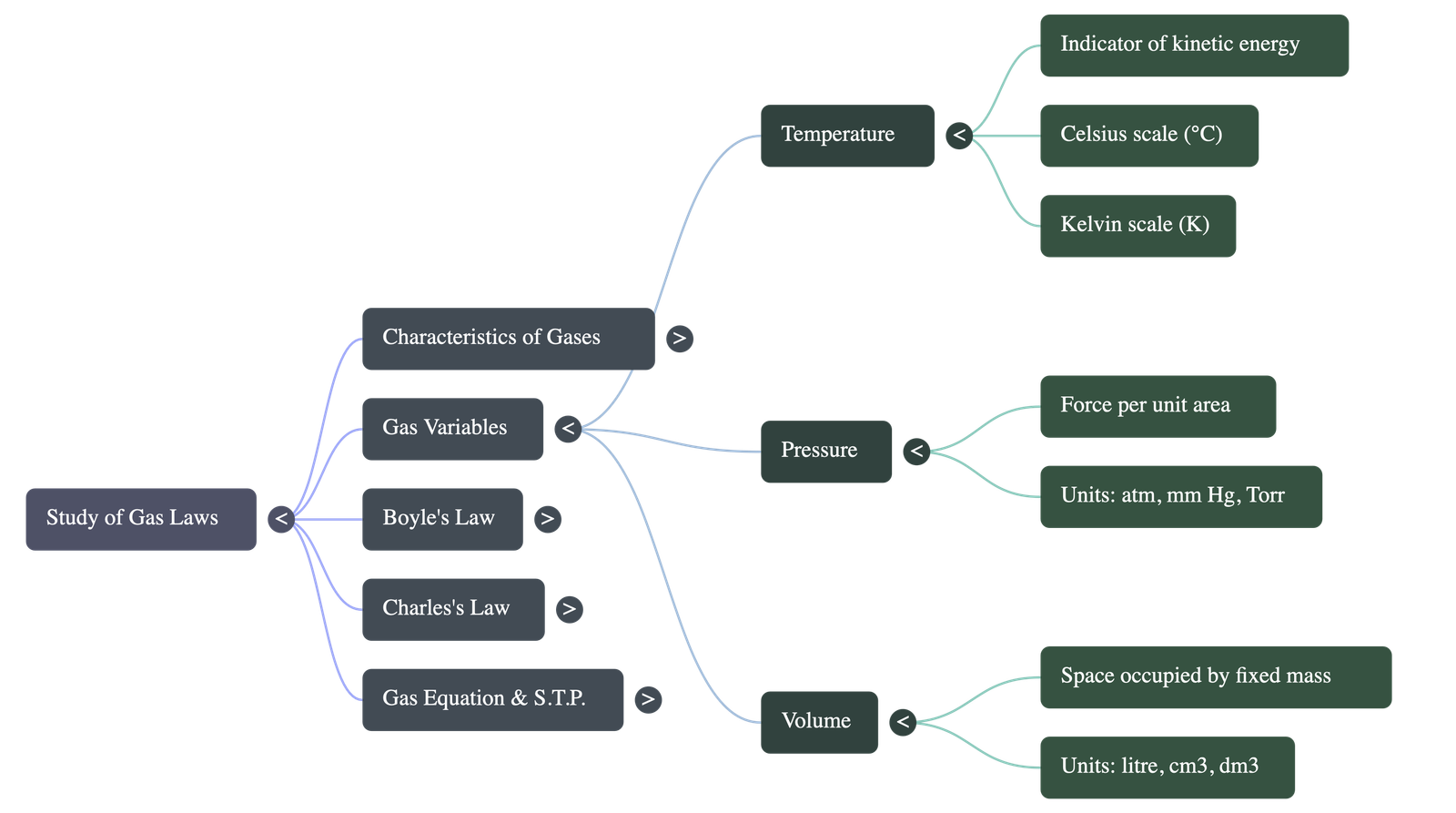
- Temperature: An indicator of the average kinetic energy possessed by molecules. Increasing temperature increases kinetic energy. Common units are Celsius and Kelvin.
- Pressure: The average force exerted by the continuous bombardment of gas molecules per unit area on the container walls. Common units are Atmospheres, cm Hg, mm Hg, and Torr.
- Volume: The three-dimensional space occupied by a fixed mass of gas. Common units are Litres, cubic decimetres, cubic centimetres, and millilitres.
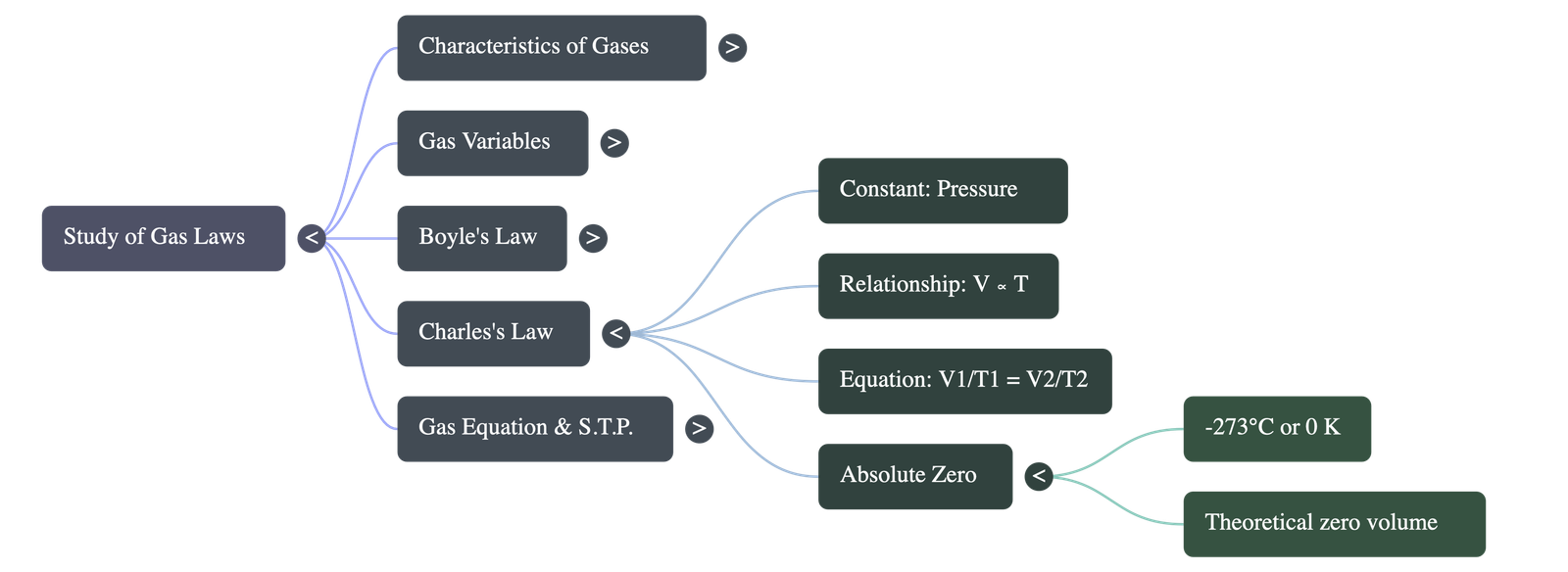
4. Boyle's Law
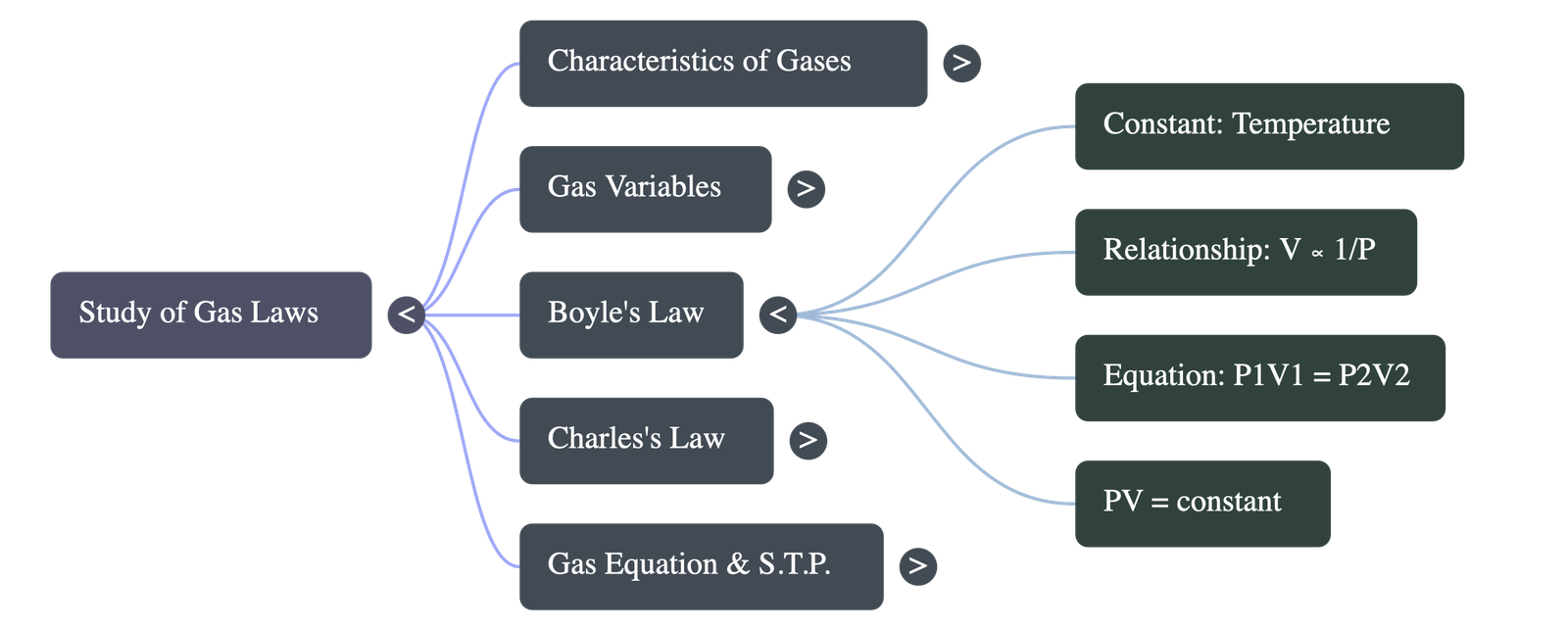
- Definition: Proposed by Robert Boyle, it states that at a constant temperature, the volume of a given mass of dry gas is inversely proportional to its pressure.
- Equation: P1 × V1 = P2 × V2 = Constant.
- Kinetic Theory Explanation: If the volume is increased at a constant temperature, molecules have to travel longer distances to strike the walls. This decreases the collision frequency, thereby lowering the overall pressure.
5. Charles' Law
- Definition: Proposed by Jacques Charles, it states that at a constant pressure, the volume of a given mass of dry gas is directly proportional to its absolute (Kelvin) temperature.
- Alternative Definition: The volume of a gas expands or contracts by 1/273 of its original volume at 0 degrees Celsius for every degree Celsius rise or fall in temperature.
- Equation: V1 / T1 = V2 / T2 = Constant.
- Absolute Zero: A theoretical temperature of -273 degrees Celsius (or 0 K) where the volume of a gas is theoretically zero. All temperatures on the Kelvin scale are strictly positive figures.
- Kinetic Theory Explanation: Doubling the absolute temperature doubles the kinetic energy and collision frequency of the molecules. To maintain a constant pressure, the volume must also double to accommodate this extra energy.
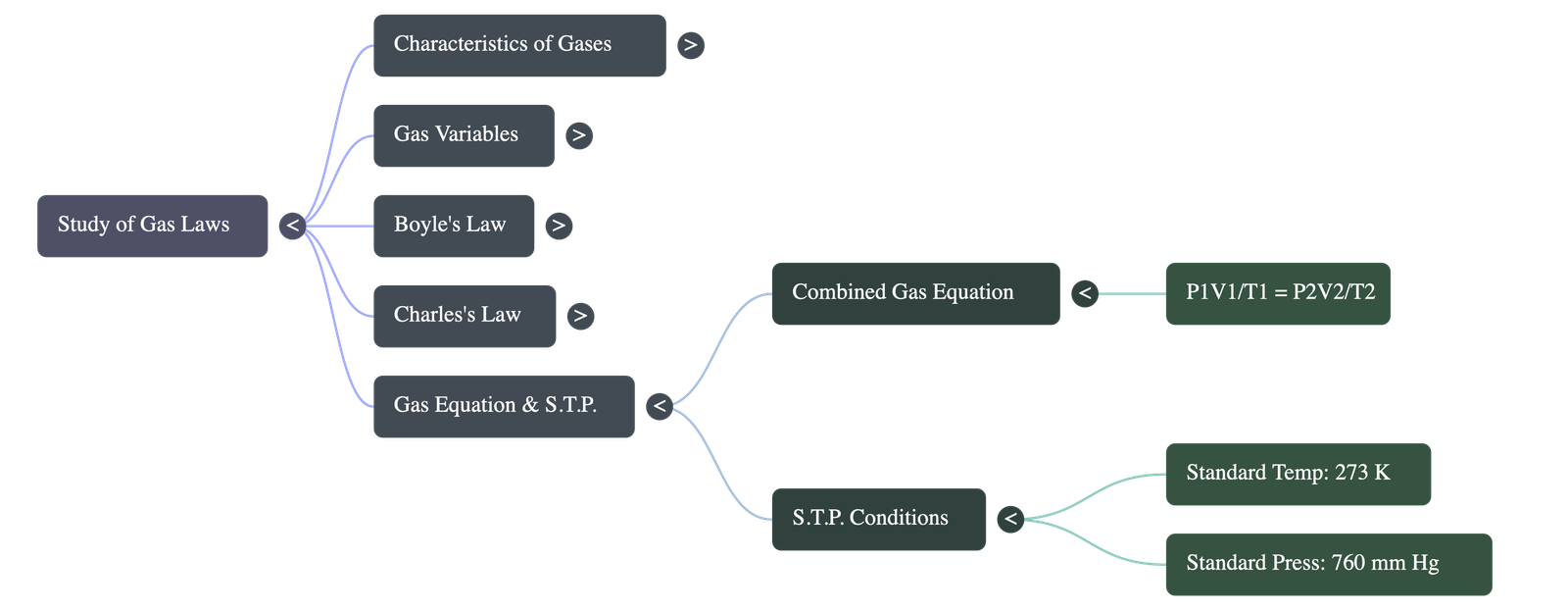
6. The Gas Equation
- Concept: An equation combining both Boyle's and Charles' laws used in chemical calculations to determine the simultaneous effects of temperature and pressure changes on volume.
- Equation: (P1 × V1) / T1 = (P2 × V2) / T2 = Constant.
7. Standard Temperature & Pressure (S.T.P.)
- Need for S.T.P.: Since gas volumes change significantly with environmental temperature and pressure variations, standard benchmark values are chosen so that different gas volumes can be easily compared.
- Standard Temperature: 0 degrees Celsius, which translates to exactly 273 Kelvin.
- Standard Pressure: 760 mm Hg (which is equivalent to 76 cm Hg or 1 atmosphere).
Quick Navigation:
| | | |
1 / 1
Quick Navigation:
| | | |