Quick Navigation:
| | | |
Atomic Structure & Chemical Bonding
1. Introduction to Atomic Theory
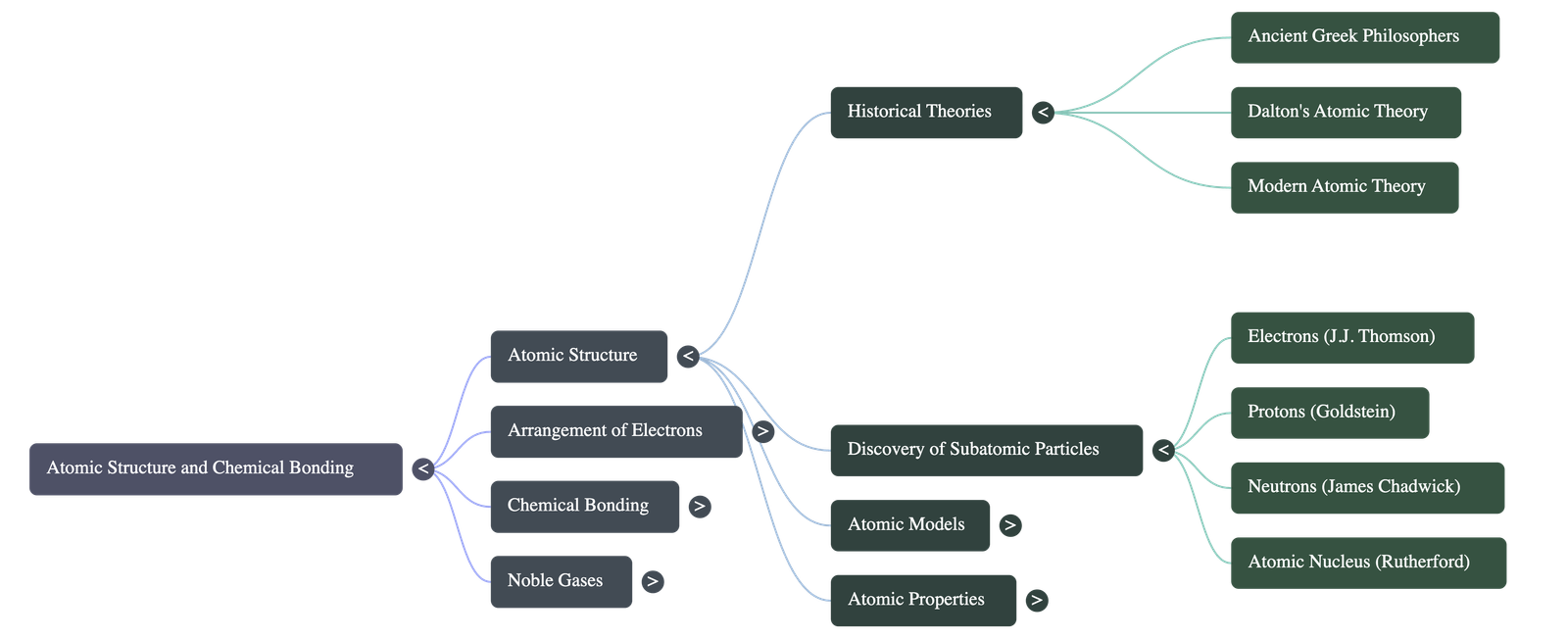
- Early Concepts: The concept of the atom originated from ancient Greek philosophers like Democritus, who studied the basic constituents of matter.
- Dalton's Atomic Theory (1808): Proposed that matter consists of small, indivisible, and indestructible particles called atoms. Atoms of the same element are identical, and atoms can neither be created nor destroyed.
- Modern Atomic Theory: Contradicts Dalton by proving atoms are divisible into subatomic particles (electrons, protons, neutrons). It also shows that atoms of the same element can have different masses (isotopes).
2. Discovery of Subatomic Particles
- Electrons: Discovered by William Crookes via cathode rays. J.J. Thomson later proved these rays travel in straight lines, possess kinetic energy, and consist of negatively charged, identical particles called electrons.
- Protons: Discovered by Goldstein through the observation of positive rays in a discharge tube. Protons are positively charged particles located in the atom.
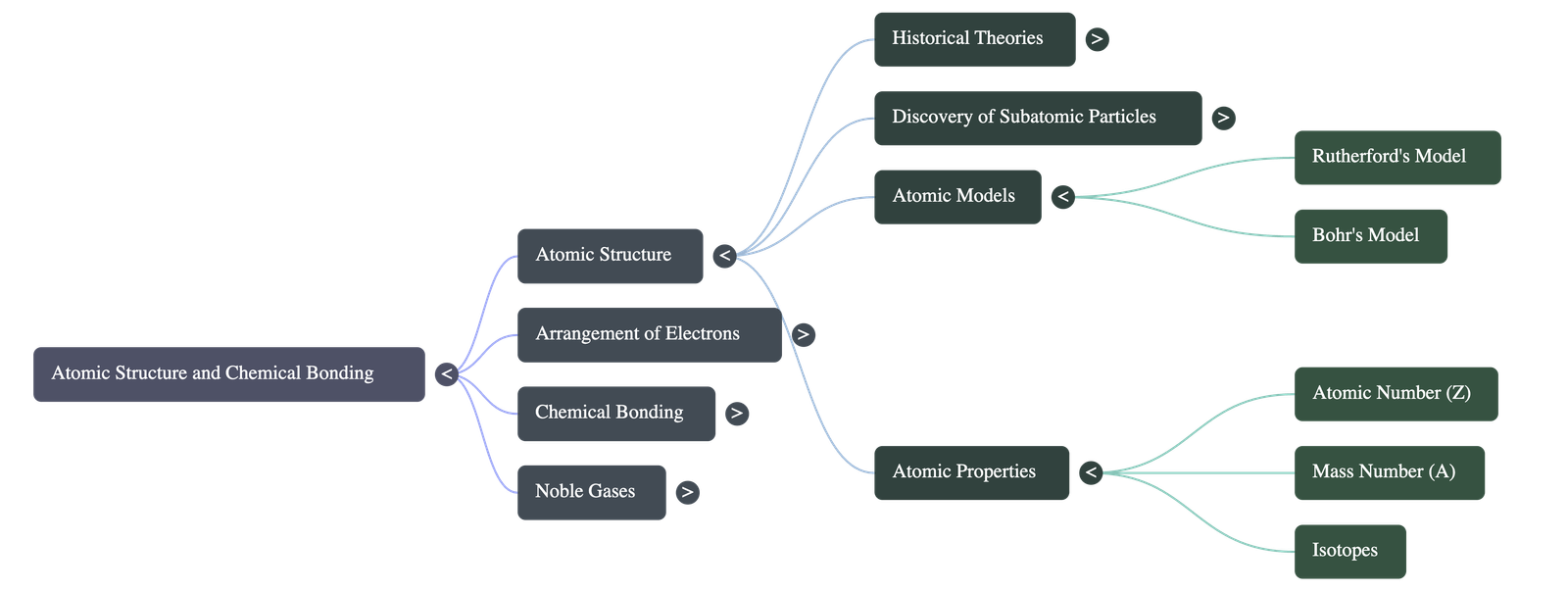
- Atomic Nucleus: Discovered by Lord Rutherford in 1911 using the alpha-particle scattering experiment on gold foil. He concluded that an atom is mostly empty space with a dense, positively charged center (the nucleus).
- Bohr's Atomic Model: Addressed the instability of Rutherford's model by proposing that electrons revolve around the nucleus in fixed orbits or energy levels without radiating energy.
- Neutrons: Discovered by James Chadwick in 1932. Neutrons carry no electrical charge but have a mass almost equal to that of a proton.
3. Representing an Atom
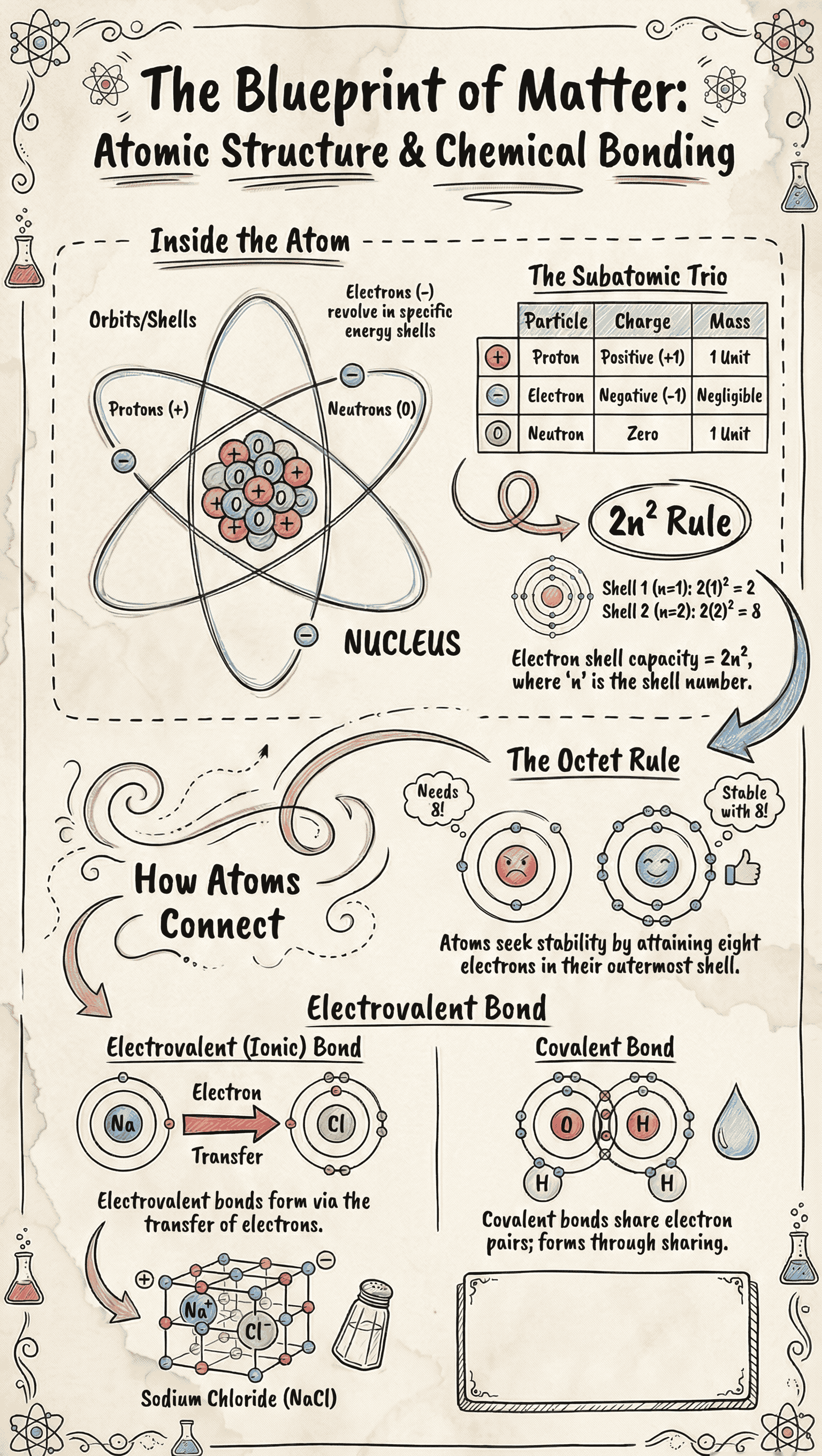
- Atomic Number (Z): The number of protons inside the nucleus. In a neutral atom, it is also equal to the number of electrons.
- Mass Number (A): The total number of protons and neutrons (collectively called nucleons) in an atom's nucleus.
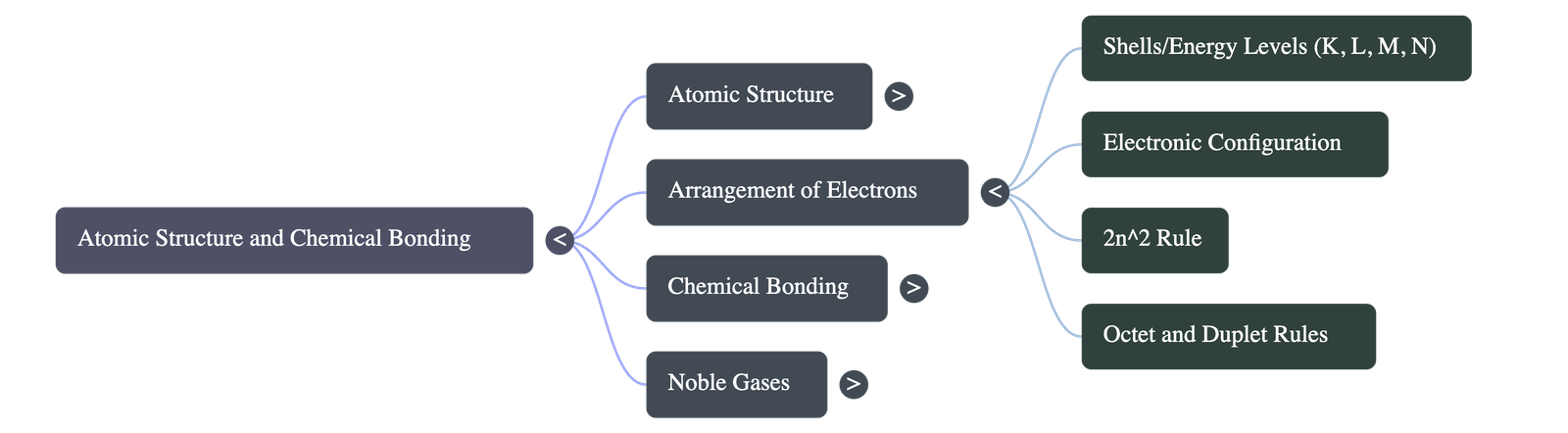
- Electron Distribution: Electrons are arranged in energy shells (K, L, M, N...). The maximum number of electrons a shell can hold is determined by the formula 2n².
- Octet Rule limit: The outermost shell of an atom cannot hold more than 8 electrons, regardless of its theoretical maximum capacity.
4. Isotopes
- Definition: Atoms of the same element that have the identical atomic number but different mass numbers due to a differing number of neutrons.
- Properties: Isotopes exhibit similar chemical properties (because their electron configurations are identical) but different physical properties (due to different atomic masses).
- Fractional Atomic Mass: Elements like chlorine exist as a mixture of isotopes in nature, which makes their average atomic mass a fraction (e.g., Chlorine has an atomic mass of 35.5).
5. Chemical Activity & The Octet Rule
- Noble Gases: Elements with completely filled outermost shells (8 electrons for the Octet rule, or 2 for Helium under the Duplet rule). They are highly stable and chemically unreactive.
- Reason for Chemical Activity: Atoms of other elements have incomplete valence shells. They chemically react by losing, gaining, or sharing electrons to achieve the stable electronic configuration of the nearest noble gas.
6. Chemical Bonding
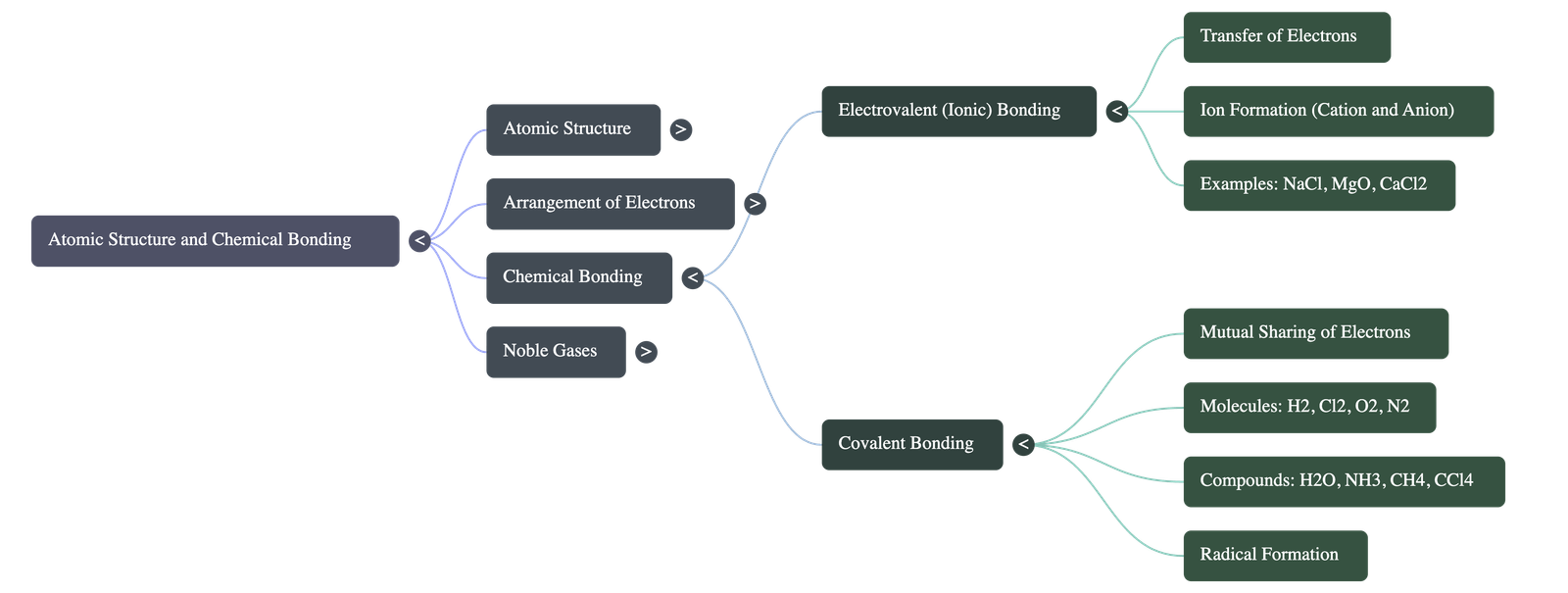
- Electrovalent (Ionic) Bonding: Formed through the complete transfer of valence electrons from a metallic atom to a non-metallic atom.
- The metal loses electrons to form a positively charged cation, while the non-metal gains electrons to form a negatively charged anion.
- These oppositely charged ions are held together by strong electrostatic forces.
- Examples: Sodium Chloride (NaCl), Calcium Oxide (CaO), and Magnesium Chloride (MgCl₂).
- Covalent Bonding: Formed by the mutual sharing of one or more pairs of electrons between two non-metallic atoms.
- Sharing one pair forms a single bond (e.g., Hydrogen, Chlorine, Water, Methane, Carbon Tetrachloride).
- Sharing two pairs forms a double bond (e.g., Oxygen).
- Sharing three pairs forms a triple bond (e.g., Nitrogen).
- Molecules like Water and Ammonia also feature lone pairs of electrons that do not participate in bonding.
- Radical Formation: Happens when a covalent bond breaks symmetrically (bond fission), leaving each separated atom with one of the shared electrons.
End of Chapter Summary
Quick Navigation:
| | | |
1 / 1
Quick Navigation:
| | | |