Quick Navigation:
| | | |
Water
1. Introduction & Occurrence of Water
- Water is a universal solvent and is considered the "elixir of life," essential for natural processes, the human body, and industrial applications.
- It exists in the free state as solid (ice, snow), liquid (river, sea, well water), and gas (water vapor, clouds).
- In the combined state, water is present in living matter (plants and animals) and in hydrated salts.
- Water is a compound made of hydrogen and oxygen combined in a fixed 2:1 ratio, and its components can only be separated by chemical means (like electrolysis).
- Natural water includes rain, spring, well, and river water. Treated water has undergone processing, such as distilled water (for qualitative analysis), pipe-borne water, and chlorinated water (for swimming pools).
2. Mixtures, Solutions, Colloids, and Suspensions
- True Solutions: Homogeneous mixtures where particles are invisible under a microscope, do not settle, and pass through filter paper and semi-permeable membranes.
- Colloidal Solutions: Heterogeneous mixtures where particles pass through filter paper but not a semi-permeable membrane, and do not settle down.
- Suspensions: Heterogeneous mixtures where particles are visible, settle down after some time, and cannot pass through filter paper.
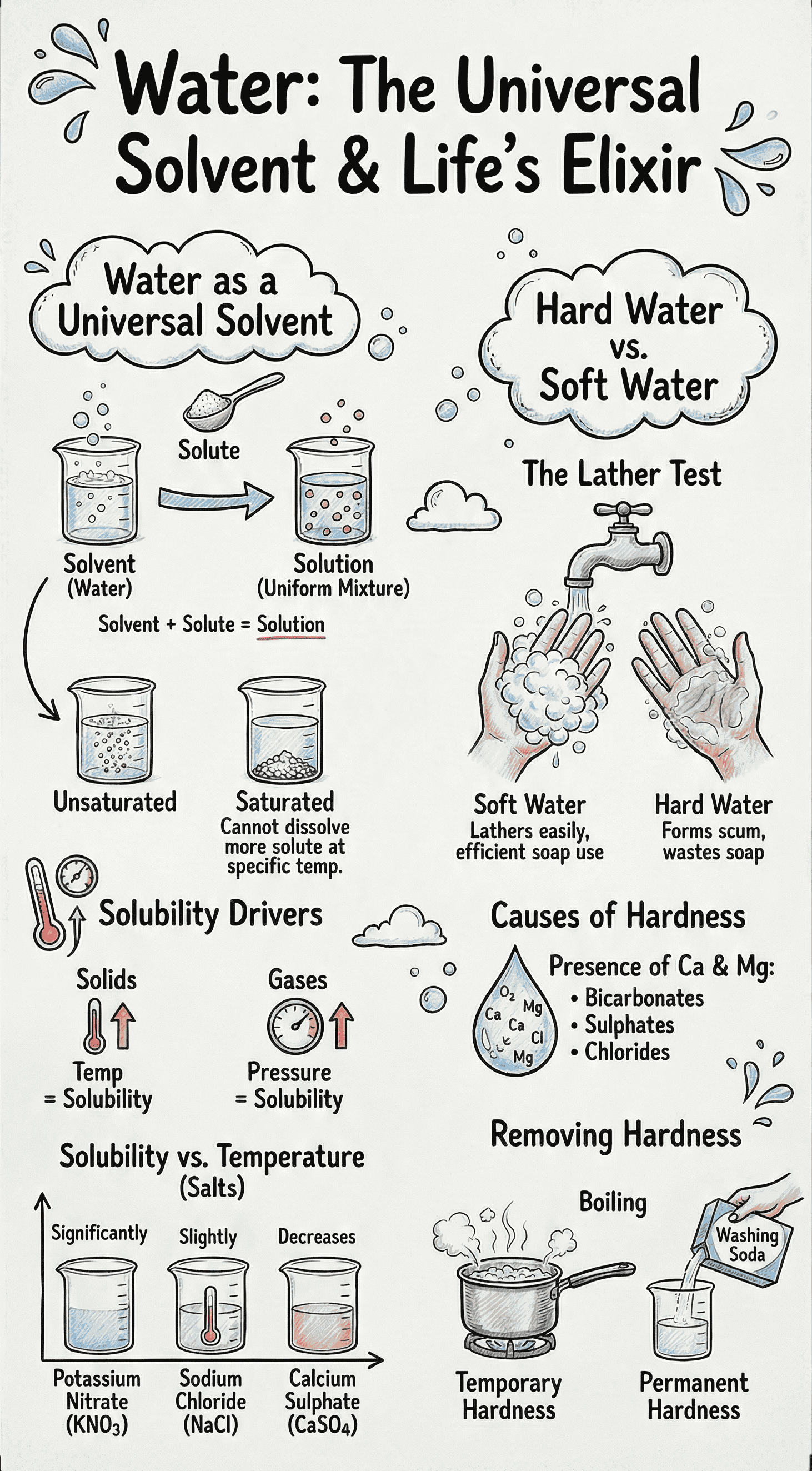
- A Solvent is the medium of dissolution (typically liquid), a Solute is the substance that dissolves, and the resulting homogeneous mixture is a Solution.
3. Types of Solutions
- Dilute vs. Concentrated: A dilute solution has a small amount of solute compared to the solvent, while a concentrated solution has a relatively large amount.
- Unsaturated Solution: Can dissolve more solute at a given temperature.
- Saturated Solution: Cannot dissolve any more solute at a given temperature. It can be made unsaturated by heating or adding more solvent.
- Supersaturated Solution: Contains more dissolved solute at a given temperature than a saturated solution could normally hold, achieved by cooling a hot saturated solution.
- Apart from water, other solvents include benzene (for rubber), turpentine oil (for paint), acetone (for nail polish), and alcohol (for iodine).
4. Solubility and Solubility Curves
- Solubility is the maximum amount of a solute in grams that will saturate 100 grams of a solvent at a particular temperature.
- A Solubility Curve graphs the effect of temperature on the solubility of a substance.
- For some solids (like potassium nitrate), solubility increases with a rise in temperature. For others (like sodium chloride), it increases only slightly.
- For some substances (like calcium sulphate), solubility decreases with a rise in temperature.
- Solubility curves help pharmacists calculate drug preparations, assist chemists in extractions, and enable the separation of solutes by crystallization.
5. Solubility of Gases
- Effect of Pressure (Henry's Law): At a given temperature, the mass of gas dissolved in a liquid is directly proportional to the pressure. Soda water fizzes when opened because the sudden decrease in pressure lowers the solubility of carbon dioxide.
- Effect of Temperature: Gas solubility in water decreases as temperature increases. This is why boiled water tastes flat (dissolved air is expelled) and why soda fizzes less when opened cold compared to when opened at room temperature.
6. Crystals, Crystallisation, and Water of Crystallisation
- Crystallisation is the separation of well-defined, geometrically shaped solid particles (crystals) from a hot saturated solution upon cooling. Slow cooling forms better, well-defined crystals compared to rapid cooling.
- Water of Crystallisation is the fixed number of water molecules that loosely combine with a substance when it crystallizes, giving the crystal its geometric shape and color.
- Hydrated substances contain this water (e.g., blue copper sulphate). Heating them drives off the water, turning them into Anhydrous substances (e.g., white, amorphous copper sulphate powder).
7. Efflorescence, Deliquescence, and Hygroscopy
- Efflorescence: Crystalline hydrated salts lose their water of crystallization to the atmosphere and turn into an amorphous powder (e.g., washing soda). It happens when the crystal's vapor pressure is higher than atmospheric humidity.
- Deliquescence: Water-soluble salts absorb moisture from the atmosphere and dissolve into a liquid state (e.g., iron(III) chloride). Common salt turns sticky in humid conditions due to deliquescent impurities like magnesium chloride.
- Hygroscopic Substances: Absorb moisture from the air but do not change their physical state (e.g., quicklime).
- Drying vs. Dehydrating Agents: Drying agents (like fused calcium chloride) absorb moisture without changing state. Dehydrating agents (like concentrated sulphuric acid) chemically remove combined water elements from compounds.
8. Hard and Soft Water
- Soft Water lathers readily with soap, while Hard Water does not, leading to soap wastage.
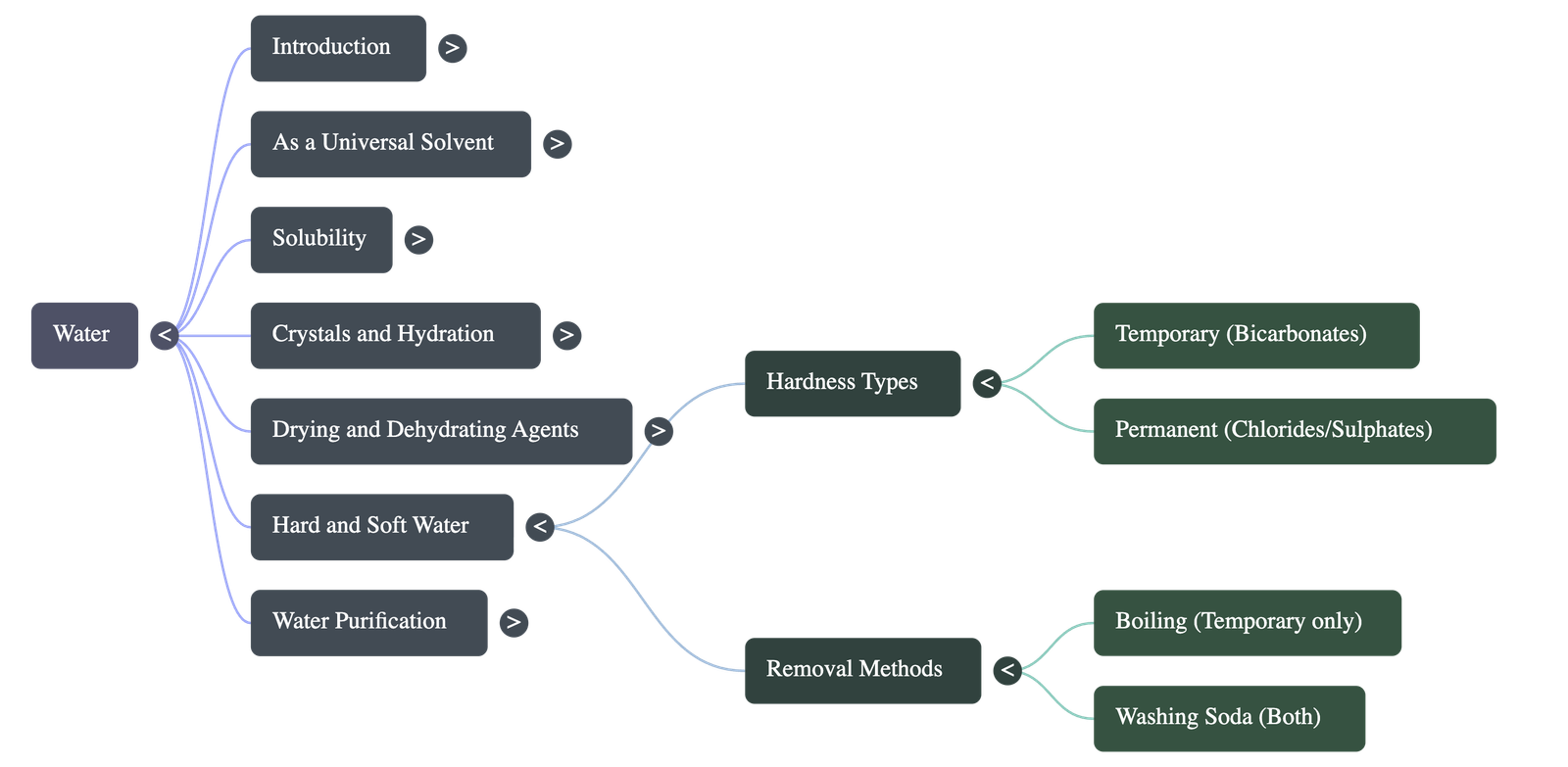
- Hardness is caused by the presence of calcium and magnesium salts (bicarbonates, sulphates, and chlorides) dissolved from the earth's surface.
- Temporary Hardness: Caused by calcium and magnesium bicarbonates. It can be removed simply by boiling, which decomposes the bicarbonates into insoluble carbonates that can be filtered out.
- Permanent Hardness: Caused by calcium and magnesium chlorides and sulphates. It cannot be removed by boiling.
- Both types of hardness can be removed by the addition of washing soda, which converts the soluble hard salts into insoluble carbonate precipitates.
- Hard water is disadvantageous as it is unfit for laundry, industrial use (causes boiler scale), and certain drinking preparations.
9. Making Water Potable
- Potable water is fit for human consumption, free from harmful bacteria and excessive minerals.
- Filtration: Passing water through sand and gravel removes suspended impurities.
- Chlorination: Adding chlorine disrupts the cell membranes of microorganisms, killing germs to prevent waterborne diseases like typhoid and cholera.
- Defluoridation: Removes excess fluoride using activated carbon to prevent dental damage and joint pains.
- Role of Alum: Acts as a coagulant. It binds suspended mud or clay particles into heavier particles that settle at the bottom, making filtration easier.
Quick Navigation:
| | | |
1 / 1
Quick Navigation:
| | | |