Quick Navigation:
| | | |
Chemical Changes & Reactions
A. Introduction to Chemical Reactions
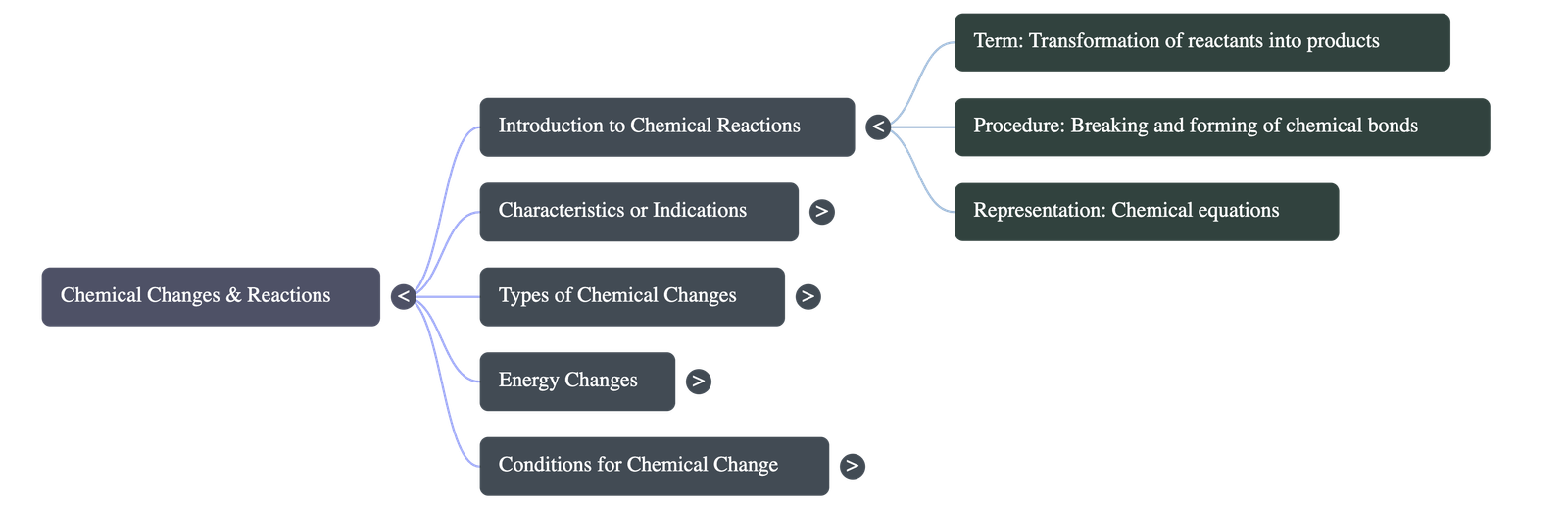
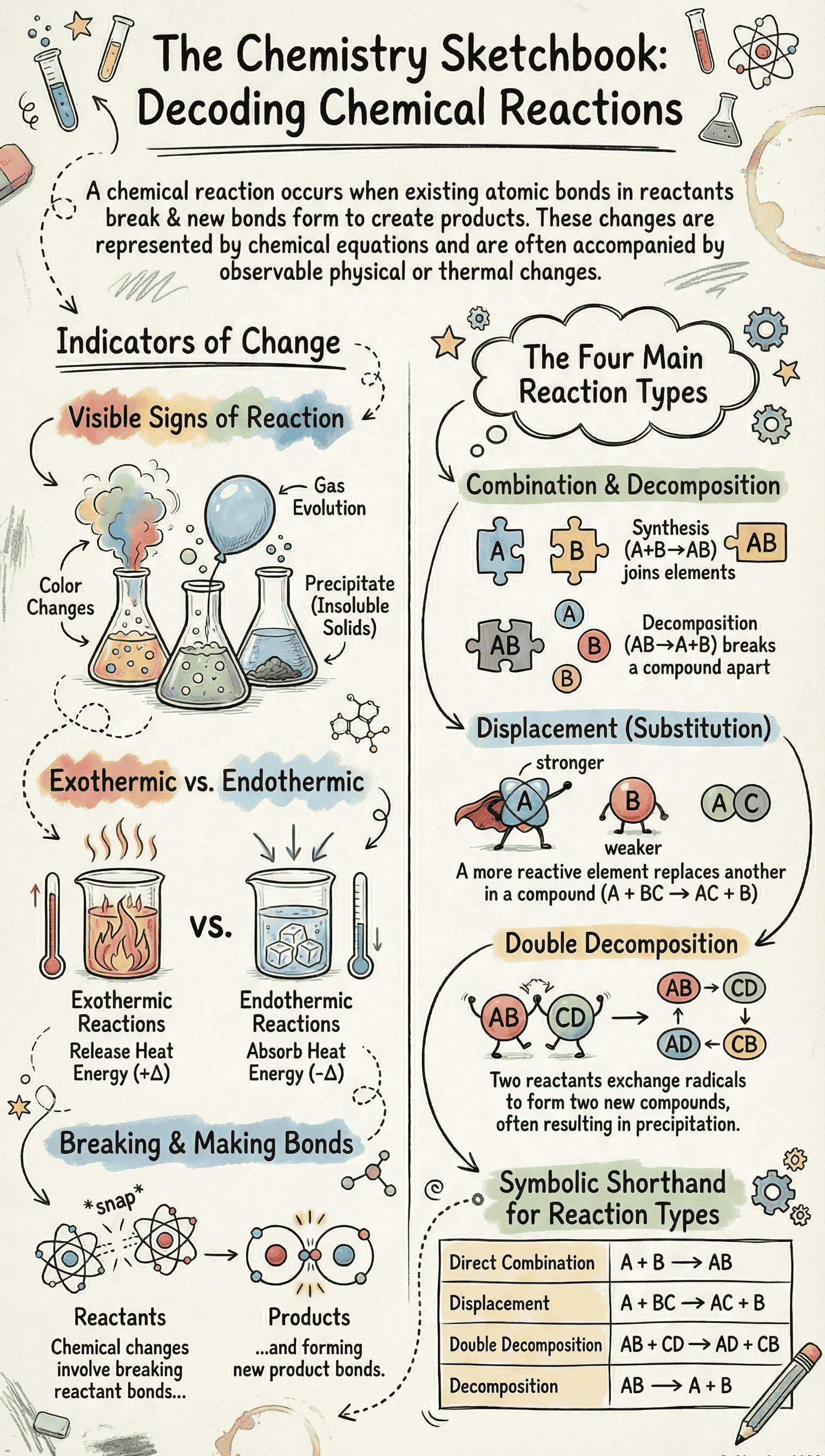
- Definition: A chemical reaction is a chemical change where matter is transformed into one or more new substances.
- The Procedure: Existing chemical bonds in the reactants are broken, and new bonds are formed to create new substances called products.
- Representation: A chemical equation acts as a shorthand form representing the result of the chemical change, showing reactants on the left-hand side and products on the right-hand side.
B. Characteristics & Indications of Chemical Reactions
Chemical reactions are often accompanied by observable external changes:
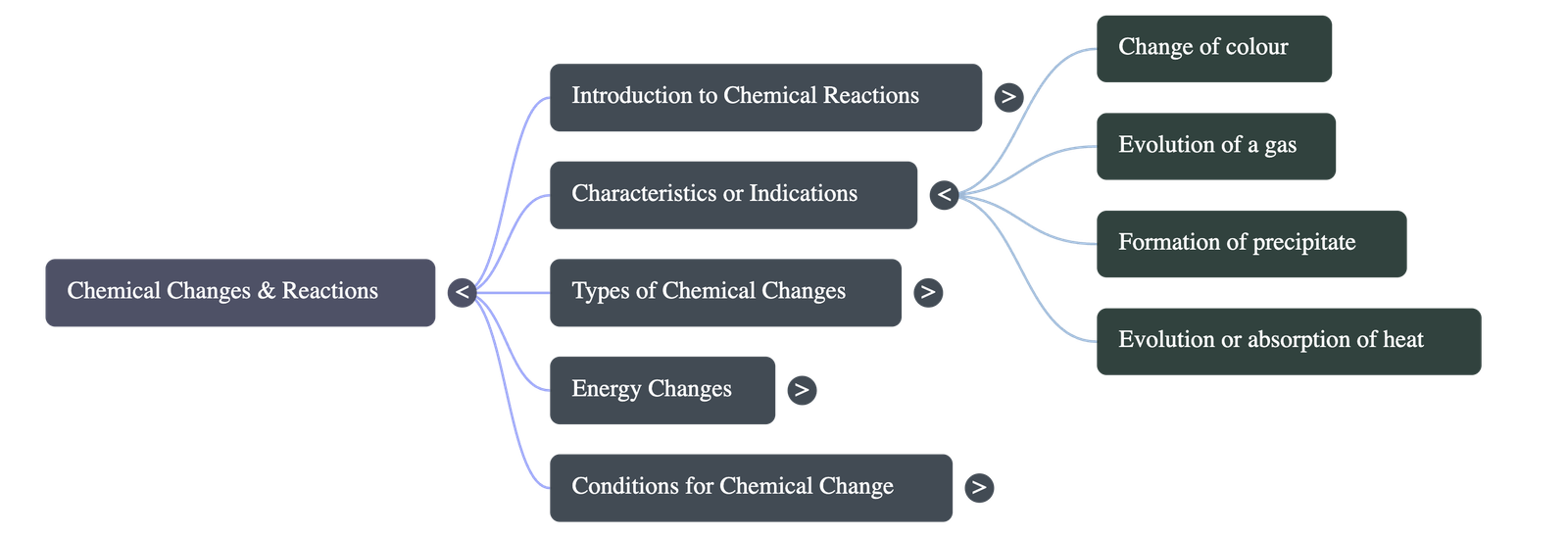
- Change of Colour: The reactants may change colour upon reacting (e.g., blue copper nitrate turns into black copper oxide upon heating).
- Evolution of a Gas: Effervescence or gas release can occur (e.g., mixing sodium sulphite with dilute sulphuric acid releases sulphur dioxide gas).
- Formation of Precipitate: Soluble reactants may form an insoluble product called a precipitate (e.g., lead nitrate and sodium chloride produce a white precipitate of lead chloride).
- Evolution or Absorption of Heat: Reactions that release heat are Exothermic, while those that absorb heat are Endothermic.
C & D. Direct Combination (Synthesis) Reactions
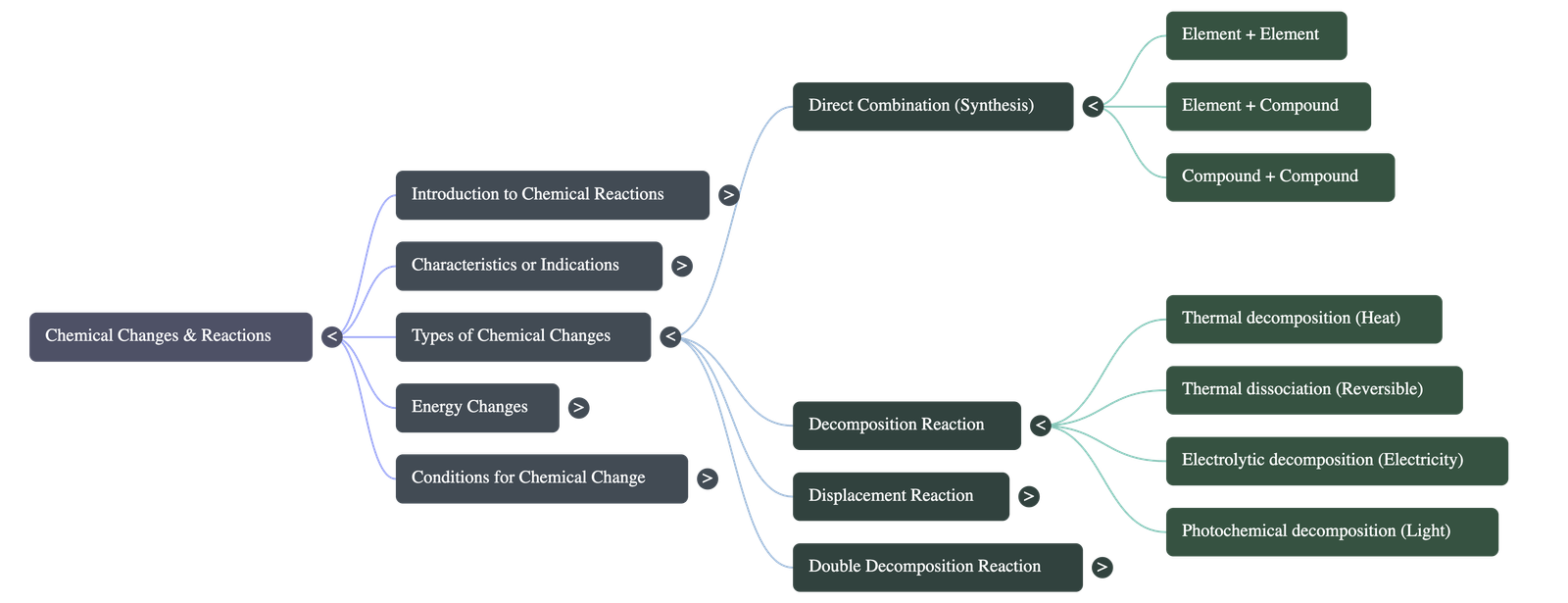
- Concept: A reaction in which two or more elements or compounds combine to form exactly one new compound.
- Types of Combinations:
- Element + Element → Compound (e.g., Carbon + Oxygen → Carbon dioxide).
- Element + Compound → New Compound.
- Compound + Compound → New Compound.
- Key Examples: Burning of hydrogen in air to give water, manufacture of ammonia from nitrogen and hydrogen, and the formation of limestone (calcium carbonate) from calcium oxide and carbon dioxide.
E. Decomposition Reactions
- Concept: A single chemical compound breaks down into two or more simpler substances.
- Thermal Decomposition: Decomposition brought about by heat. Metal oxides, hydroxides, carbonates, and nitrates decompose differently based on the metal's stability.
- Thermal Dissociation: A reversible decomposition reaction where a substance dissociates into simpler substances upon heating (e.g., ammonium chloride breaks into ammonia and hydrogen chloride).
- Electrolytic & Photochemical Decomposition: Decomposition caused by the passage of an electric current (e.g., water breaking into hydrogen and oxygen) or by light (e.g., silver salts breaking down).
- Application in Metallurgy: Concentrated metal ores (like carbonates) are heated (calcination) to convert them into metal oxides, which are easier to reduce.
F. Displacement (Substitution) Reactions
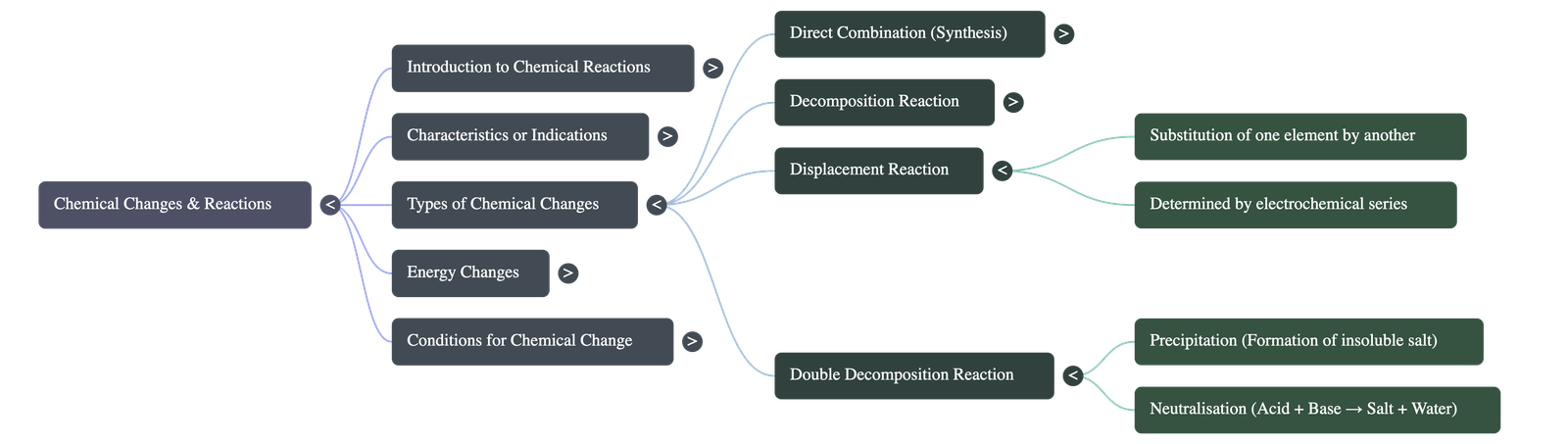
- Concept: A reaction where an element (or radical) replaces another element within a compound.
- Mechanism: A more electropositive metal (or more electronegative non-metal) displaces a less electropositive (or less electronegative) one from its aqueous solution.
- Active Metals: Metals placed above hydrogen in the electrochemical series (like magnesium, zinc, iron) can displace hydrogen from dilute acids to prepare soluble salts.
G & H. Double Decomposition Reactions
- Concept: Two reactant compounds decompose to form two new substances by mutually exchanging their radicals.
- Precipitation Reactions: A reaction between two aqueous solutions that yields at least one insoluble compound (the precipitate). This is heavily used in analytical chemistry to identify positive radicals based on the precipitate's colour.
- Neutralization Reactions: A specific double decomposition reaction between an acid and a base to produce salt and water only. Practical applications include treating acidic bee stings with basic baking soda, or using antacids to neutralize excess stomach acid.
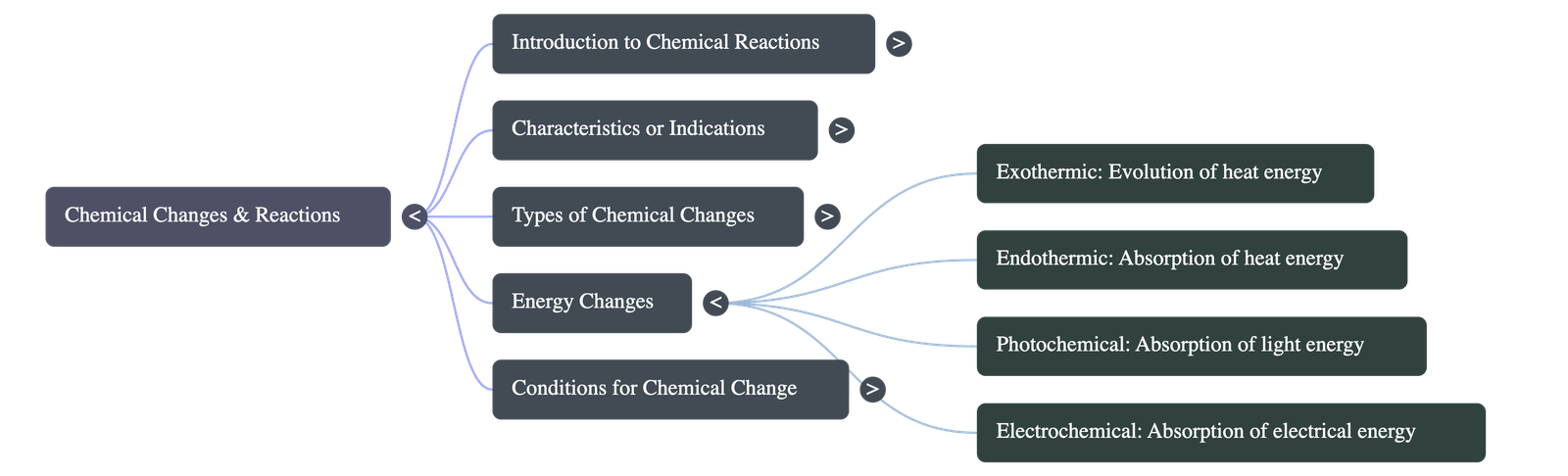
I. Energy Changes in Chemical Reactions
- Chemical Energy: Every substance possesses stored potential or latent energy. Reactions involve breaking old bonds (requires energy) and forming new bonds (releases energy).
- Exothermic vs Endothermic: If surplus energy is released, the reaction is exothermic (+ΔT). If energy is absorbed, it is endothermic (-ΔT).
- Energy Types: Energy changes can occur in the form of heat, light (Photochemical reactions like photosynthesis), or electricity (Electrochemical reactions).
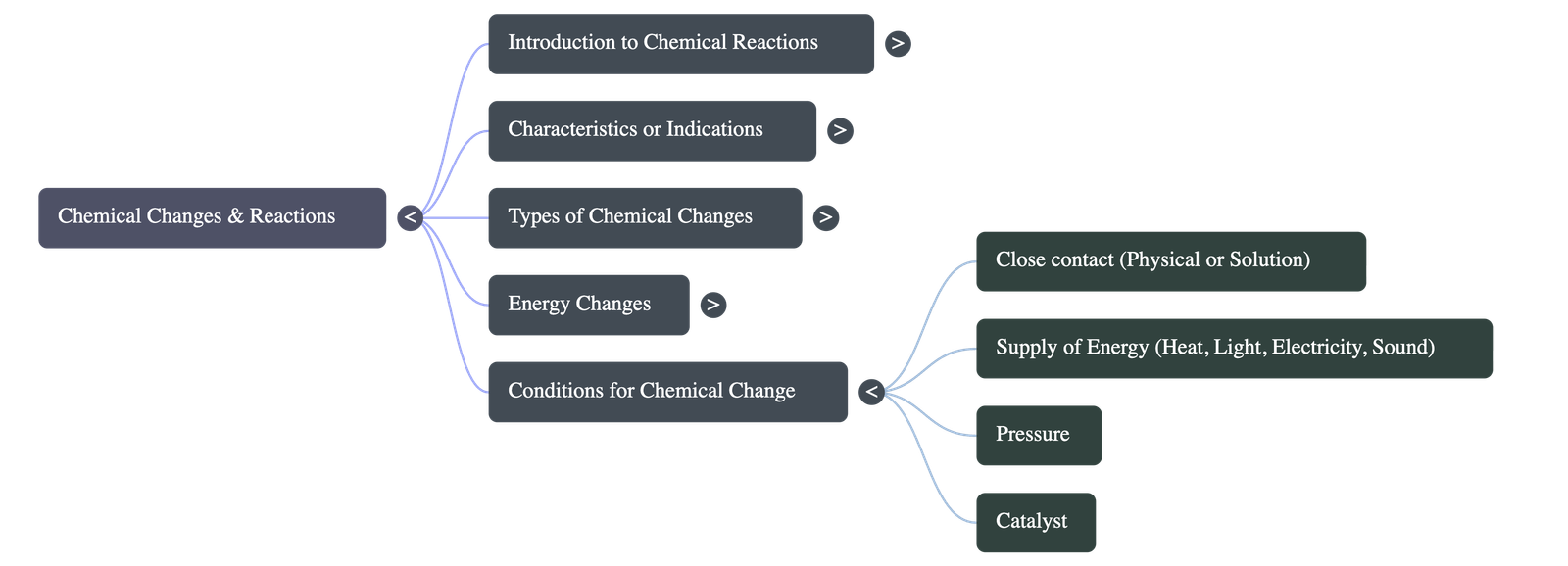
J. Conditions Required for Chemical Changes
Chemical reactions take place due to particle collisions. Specific conditions are often necessary to initiate these reactions:
- Close Contact: Reactions can be aided by physical contact (like grinding solids together) or by dissolving reactants into a solution state to increase particle mobility.
- Heat: Heating molecules makes them move faster and collide more frequently.
- Light: Some reactions require light energy to activate reactants (e.g., hydrogen and chlorine reacting in sunlight).
- Electricity: Passing an electric current directs charged particles to opposite electrodes.
- Pressure: Subjecting reactants to high pressure forces molecules to agglomerate.
- Catalyst: Use of specific substances that alter the rate of a reaction without undergoing a permanent chemical change themselves.
- Sound: Sound energy can speed up reacting molecules or ions, triggering a chemical reaction in some specific cases.
Quick Navigation:
| | | |
1 / 1
Quick Navigation:
| | | |