Quick Navigation:
| | |
The Language of Chemistry
1. Symbols of Elements
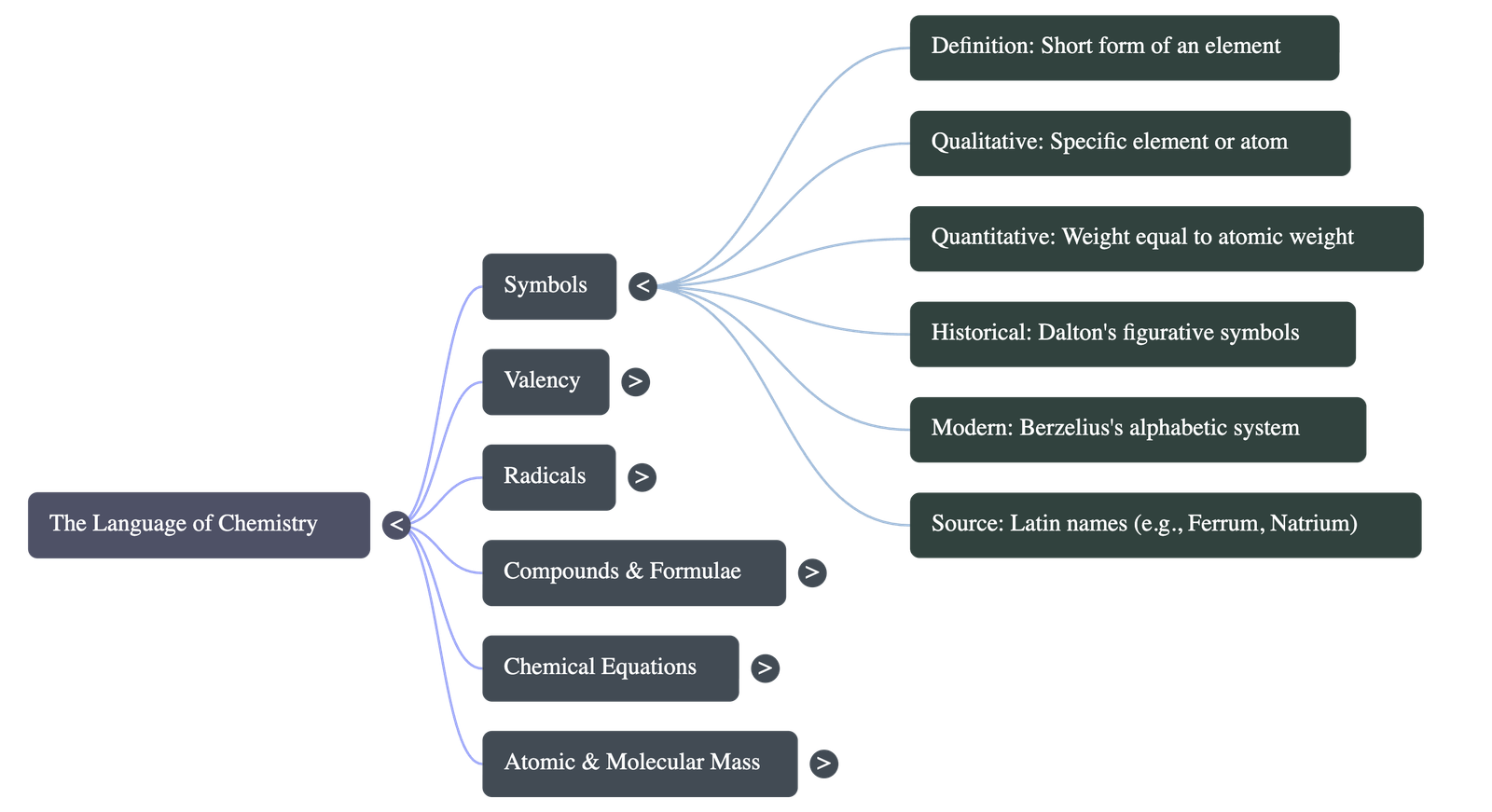
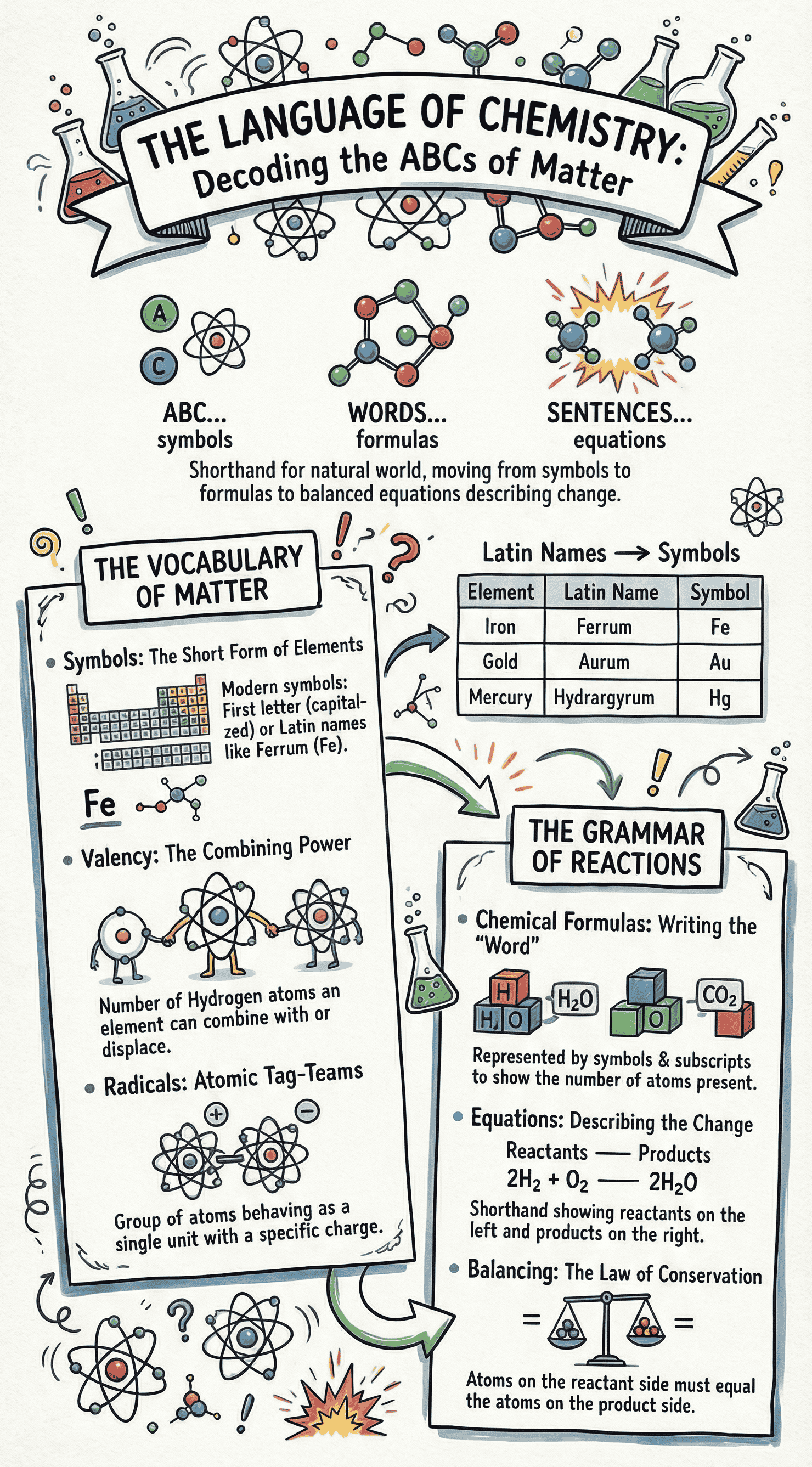
- Definition: A symbol is a short form representation of an element. It signifies a specific element, one atom of that element, and the atomic weight of the element relative to carbon-12.
- Historical Context: John Dalton (1807) initially used figurative symbols, which were later abandoned for being cumbersome. J.J. Berzelius (1814) introduced the modern system.
- Naming Conventions:
- Often uses the first letter of the element's name in capitals (e.g., C for Carbon).
- If the first letter is taken, a second small letter is added (e.g., Ca for Calcium).
- Some symbols are derived from their Latin names (e.g., Cuprum [Cu] for Copper, Ferrum [Fe] for Iron, Natrium [Na] for Sodium).
2. Valency & Radicals
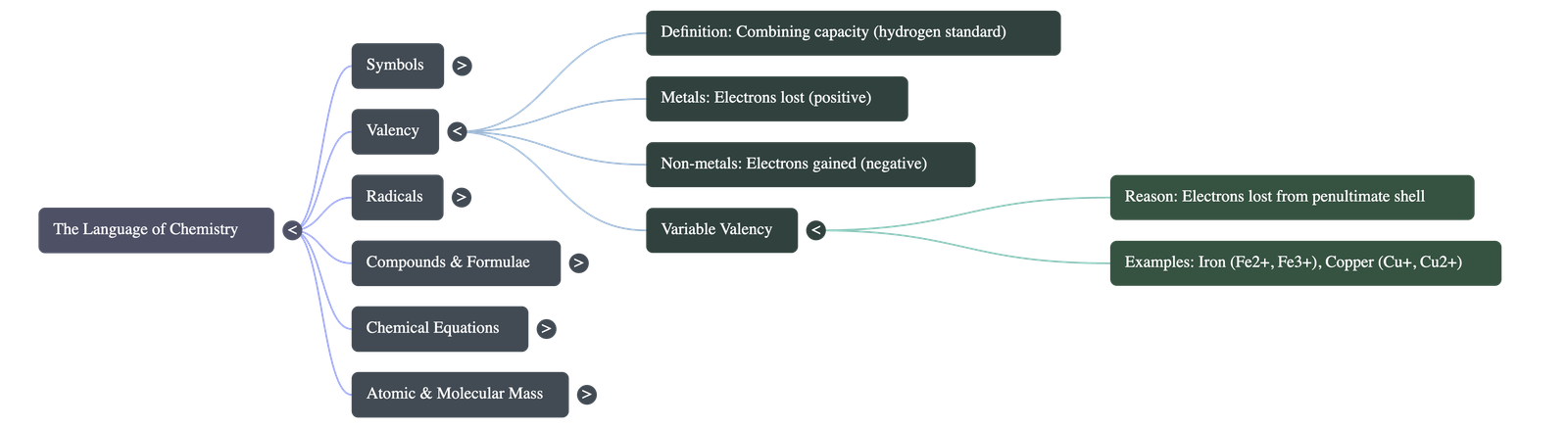
- Valency: The combining capacity of an element, defined by the number of hydrogen atoms it can combine with or displace. Metals lose electrons (positive valency), and non-metals gain electrons (negative valency).
- Variable Valency: Some metals lose electrons not only from their outermost shell but also from the penultimate (second last) shell, causing them to exhibit multiple valencies (e.g., Copper can be Cu1+ or Cu2+).
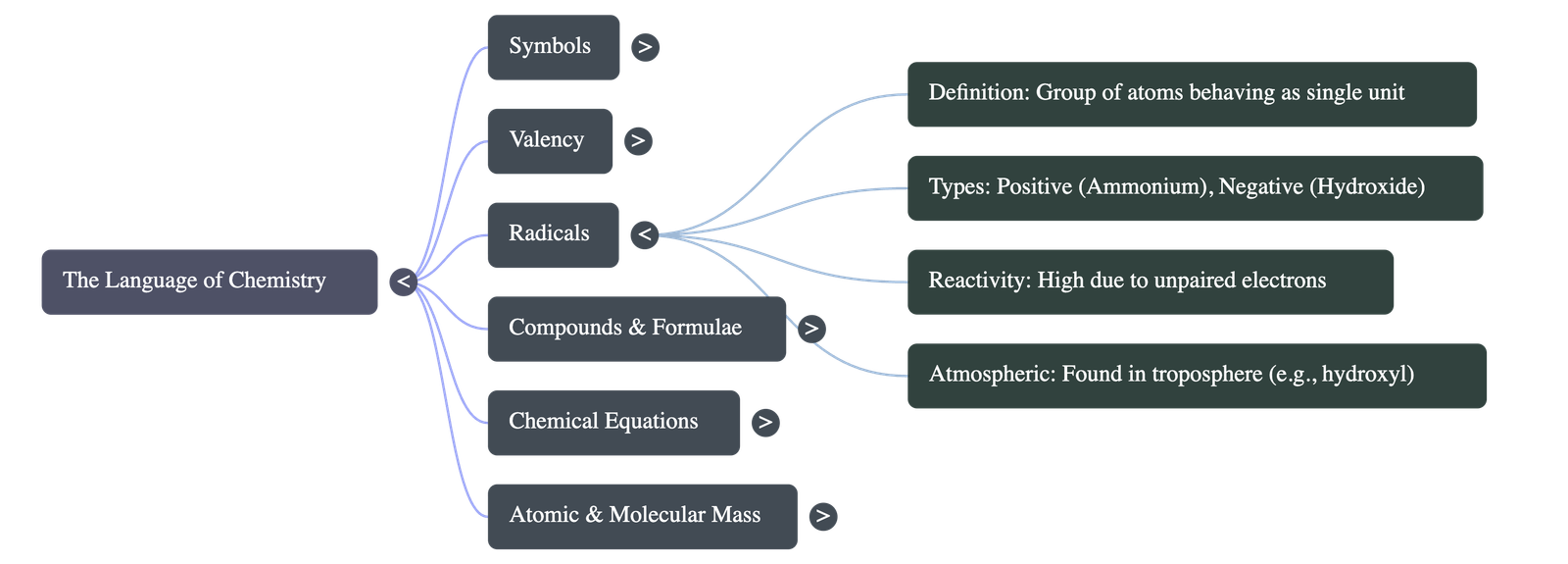
- Radicals: An atom or a group of atoms behaving as a single unit with a specific charge and valency (e.g., Ammonium NH41+). They contain unpaired valence electrons, making them highly reactive.
- Atmospheric Radicals: Formed in the lower troposphere by the breaking of chemical bonds via sunlight. They react with pollutants and can cause respiratory issues but are eliminated by rain or chemical oxidation.
3. Chemical Compounds & Formulas
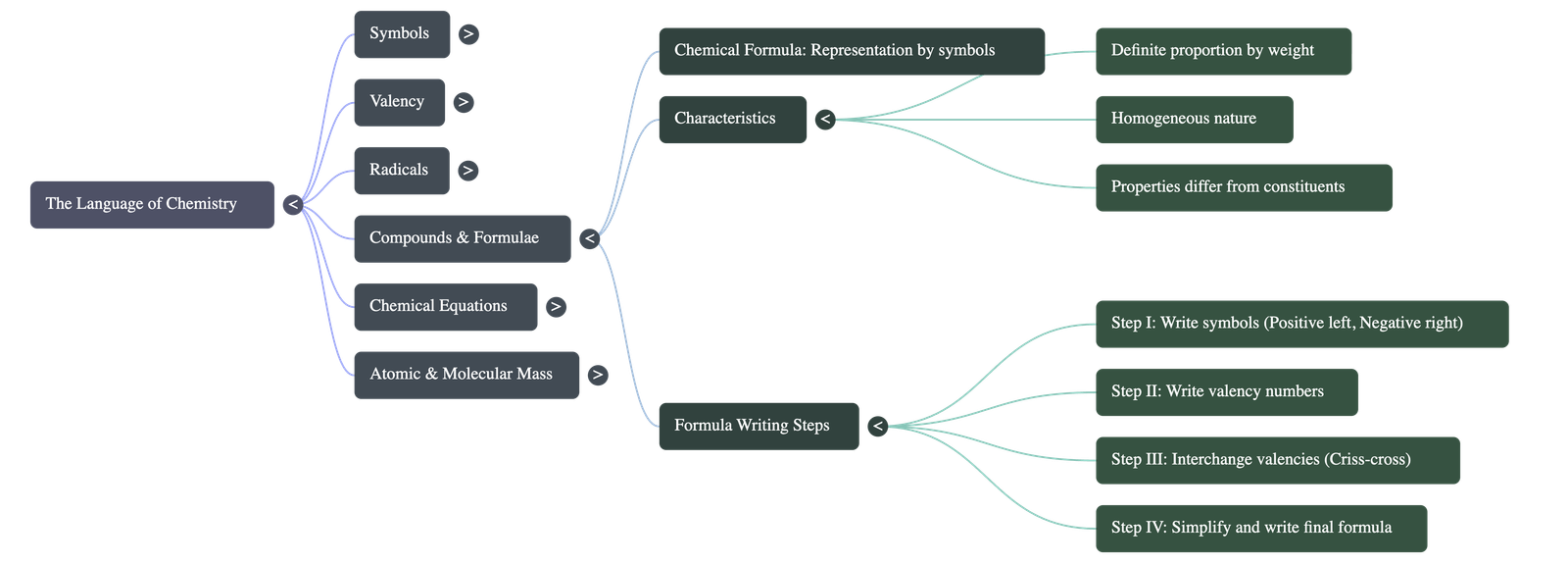
- Compound Definition: A pure substance composed of two or more elements chemically combined in a fixed proportion by weight.
- Characteristics: Compounds are homogeneous, possess a definite set of properties distinct from their constituent elements, and their components can only be separated by chemical means.
- Key Classes of Compounds: The chapter outlines specific formulas for Acids, Bases, Salts, Hydrocarbons (organic compounds containing only carbon and hydrogen), and Alcohols.
- Writing Formulas:
- Write the symbol of the positive atom/radical on the left and the negative on the right.
- Write the valency number on the top right.
- Interchange the valency numbers and shift them to the lower right. Cancel out common factors to achieve a simple ratio.
- Naming Rules: Metals are written first with non-metals given an '-ide' suffix. Compounds with two non-metals use prefixes (mono-, di-, tri-). When oxygen is present, suffixes like '-ate' (more oxygen) or '-ite' (less oxygen) are used.
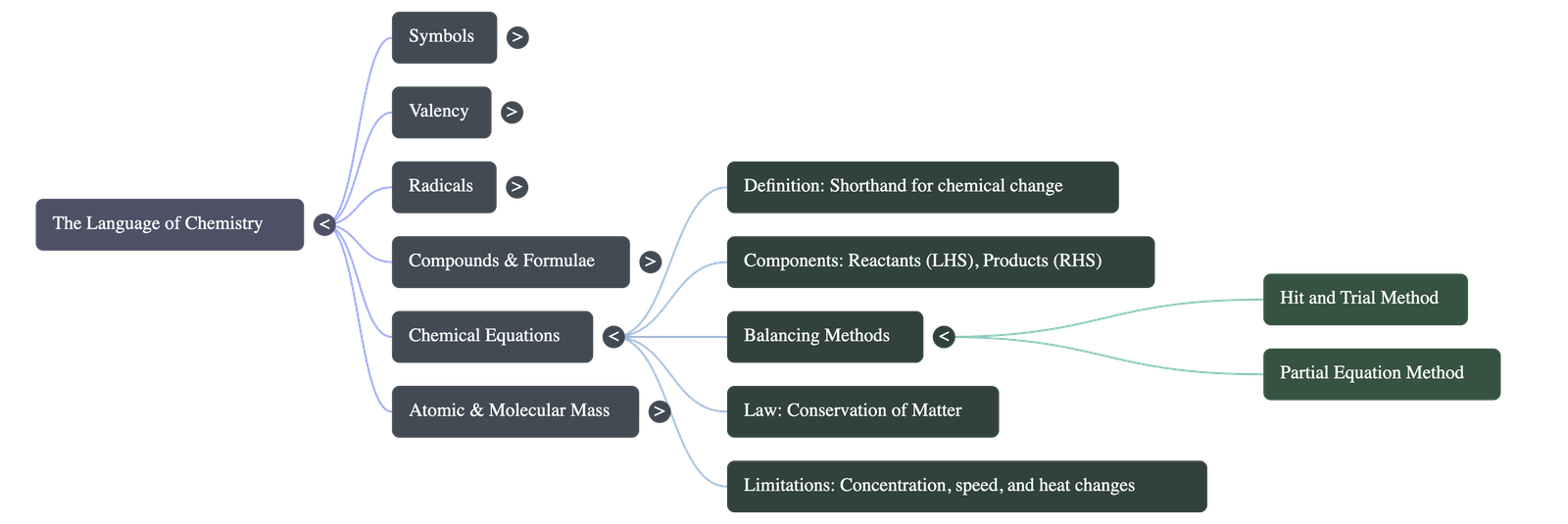
4. Chemical Equations
- Definition: A shorthand representation of a chemical change, showcasing reactants (substances taking part) on the left and products (substances formed) on the right.
- Information Provided: Shows the chemical nature of reactants/products, the direction of the reaction, and the physical state (solid, liquid, gas).
- Limitations: A standard equation does not show the concentration of substances, the speed of the reaction, color changes, light/sound energy changes, or whether the reaction goes to completion.
- Balancing Equations: Equations must be balanced to obey the 'Law of Conservation of Matter' (matter is neither created nor destroyed).
- Hit & Trial Method: Counting and equalizing the number of atoms of each element on both sides.
- Partial Equation Method: Assuming a complex reaction happens in steps, balancing individual steps, and adding them together while canceling common terms.
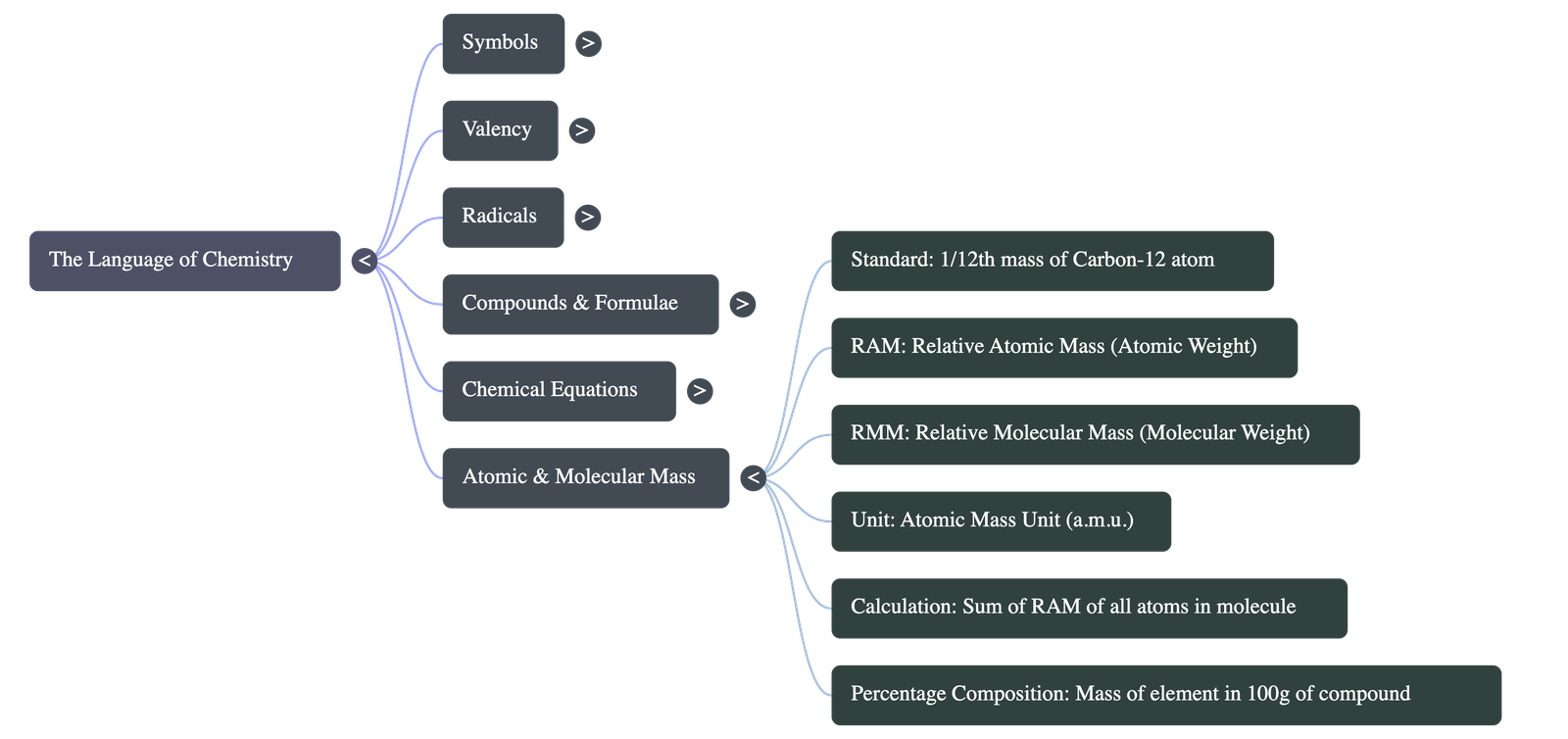
5. Relative Atomic & Molecular Mass
- Relative Atomic Mass (RAM): The number of times one atom of an element is heavier than 1/12th the mass of an atom of carbon-12. Expressed in atomic mass units (a.m.u.).
- Relative Molecular Mass (RMM): The number of times one molecule of a substance is heavier than 1/12th the mass of a carbon-12 atom.
- Fractional Weights: Atomic weights are often not whole numbers because natural elements exist as mixtures of isotopes. The atomic weight is the weighted average of these natural isotopes.
- Calculations:
- Molecular Mass: Calculated by adding together the atomic weights of all the atoms present in a molecule.
- Percentage Composition: Calculated by finding the total weight of a specific element in the compound, dividing it by the total molecular weight of the compound, and multiplying by 100.
Quick Navigation:
| | |
1 / 1
Quick Navigation:
| | |