Quick Navigation:
| | | |
Atmospheric & Environmental Chemistry
Introduction to Atmospheric Pollution
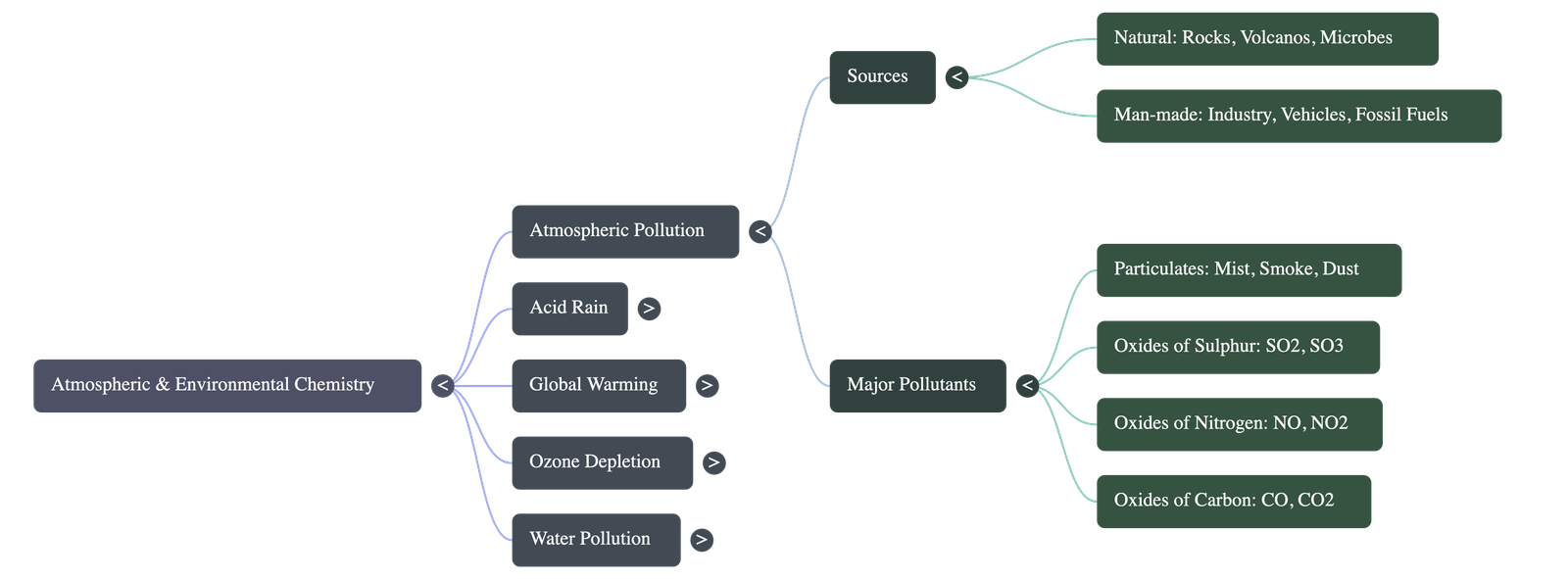
- Definition: Atmospheric pollution refers to conditions where foreign elements (from natural or man-made sources) make the air unclean, causing adverse effects on living organisms.
- Key Pollutants:
- Particulates: Mist, smoke, fumes, and dust.
- Oxides of Sulphur: Mainly sulphur dioxide, produced by burning fossil fuels and volcanic eruptions.
- Oxides of Nitrogen: Produced by automobile exhausts, high temperatures in furnaces, and lightning.
- Oxides of Carbon: Carbon monoxide and carbon dioxide from incomplete and complete combustion of fossil fuels respectively.
- Hydrogen Sulphide: Released from the decay of plants and animals, and volcanic eruptions.
- Major Consequences: These pollutants primarily lead to Acid Rain, Global Warming, and Ozone Destruction.
Acid Rain
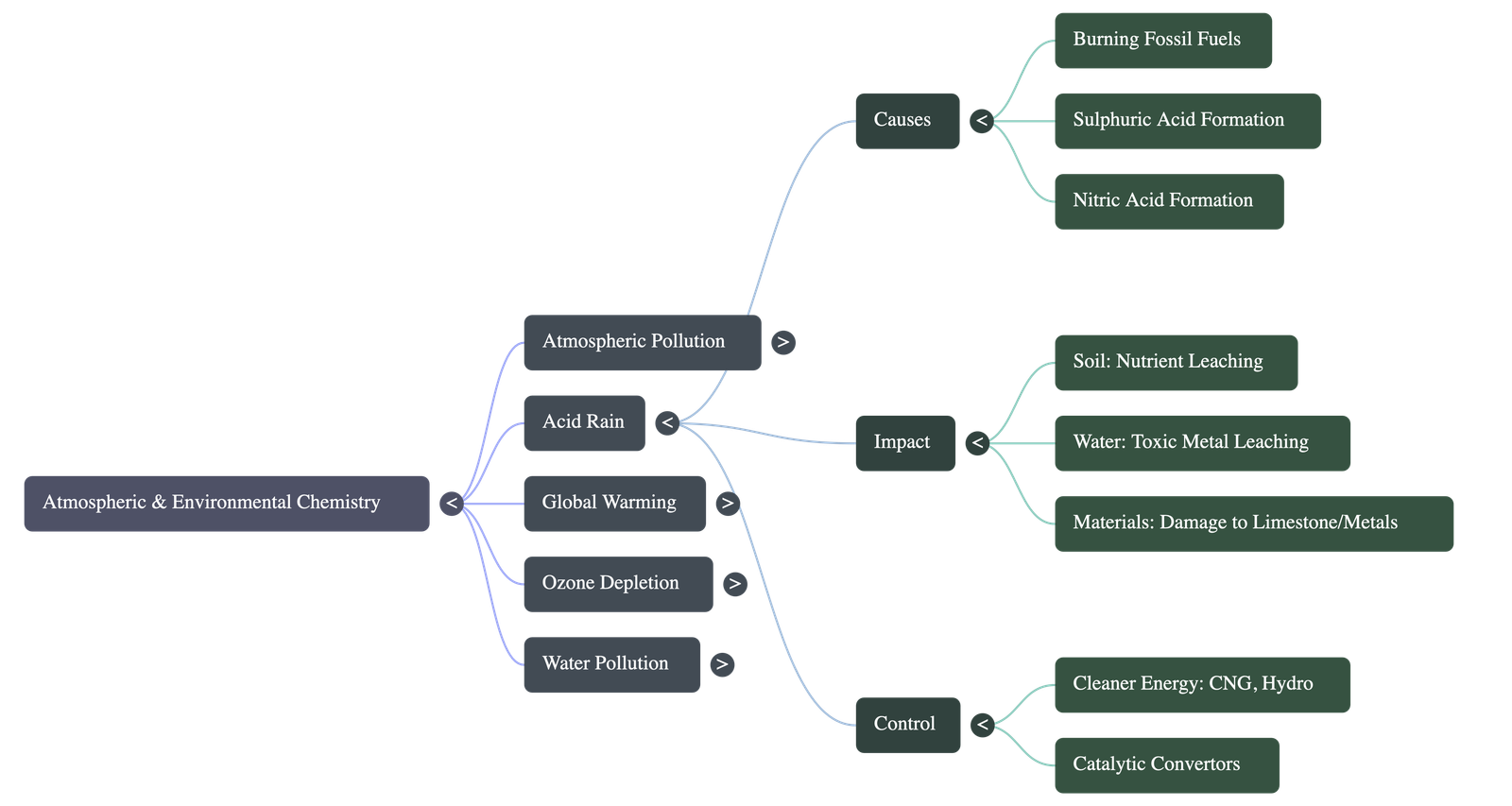
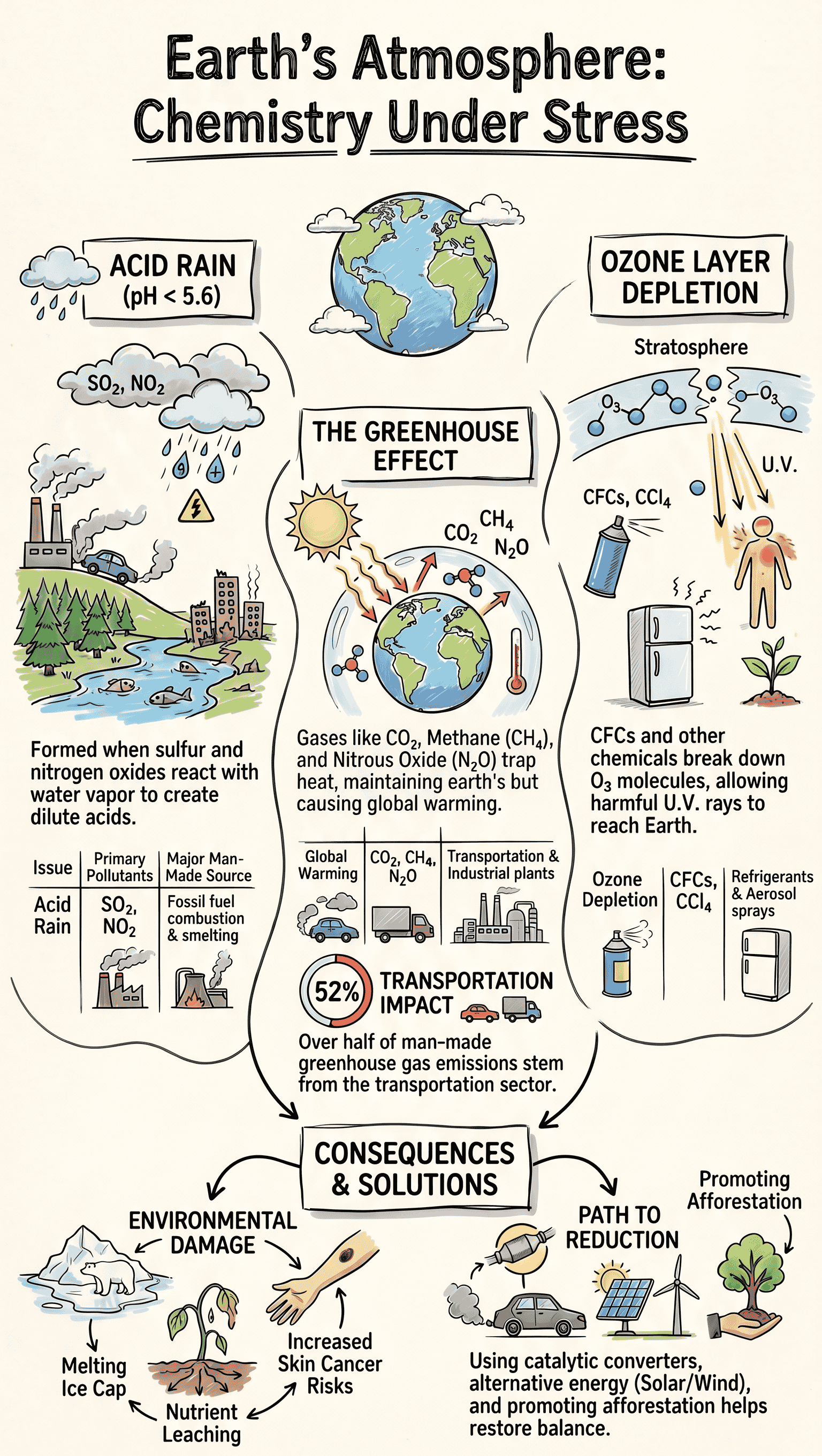
- What is it? Rain or any precipitation with a pH level less than 5.6. Pure water has a pH of 7, and unpolluted natural rain is slightly acidic (pH 5 to 6) due to dissolved carbon dioxide.
- Forms of Deposition: Occurs as wet deposits (rain, snow, fog) and dry deposits (particles containing sulphates and nitrates).
- Primary Causes: Oxides of sulphur and nitrogen interact with water vapour and sunlight to form a mist of sulphuric acid and nitric acid, which eventually falls as acid rain.
- Impact on Soil: Increases soil acidity and leaches essential nutrients like potassium and calcium. It also reduces soil fertility by inhibiting nitrogen-fixing bacteria in leguminous plants.
- Impact on Water Bodies: Lowers the pH of aquatic environments (lethal to marine life if pH drops below 4) and leaches toxic metals like mercury, lead, and zinc into streams.
- General Impact: Retards plant growth, enters the human food chain causing toxicity, and corrodes building materials and monuments (such as statues made of marble/limestone).
- Control Measures: Utilizing alternative, cleaner energy sources (CNG, wind, hydro power) and using technical devices like catalytic converters in automobiles to reduce nitrogen oxide emissions.
Global Warming & The Greenhouse Effect
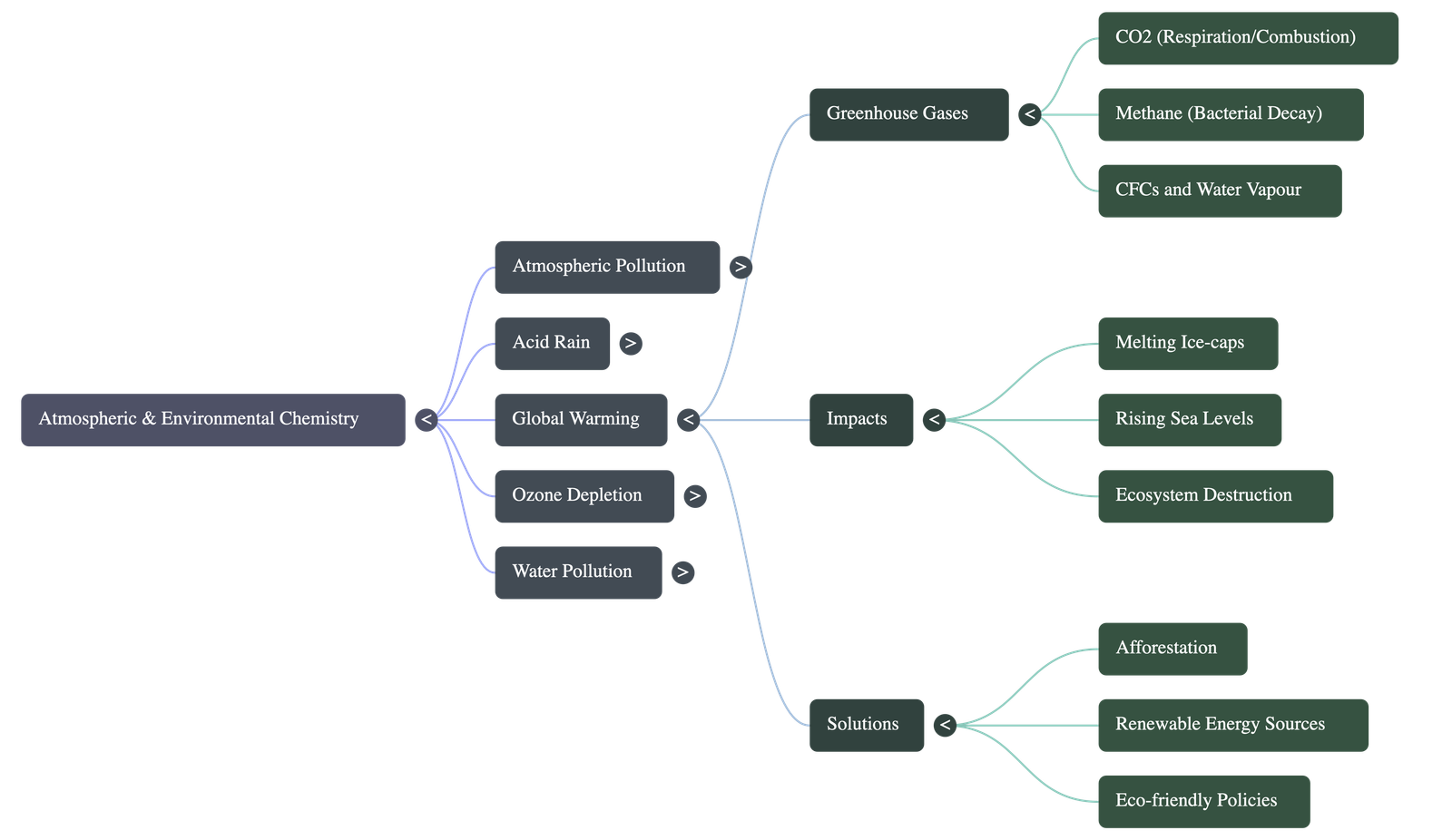
- Greenhouse Gases: Gases that effectively trap heat in the atmosphere, including carbon dioxide (major contributor at 55%), methane, water vapour, nitrous oxide, and chlorofluorocarbons (CFCs).
- Mechanism: Short-wave solar radiation passes through the atmosphere, but the long-wave heat radiation reflected back from the earth is trapped by the greenhouse gases. This blanketing effect increases the earth's surface temperature.
- Sources of Gases:
- Carbon Dioxide: Fossil fuel combustion, respiration, deforestation, and industrial activities.
- Methane: Bacterial decay in wetlands, rice paddies, coal mining, and agricultural activities.
- CFCs: Chemical coolants in air conditioners, refrigerators, and cleaning solvents.
- Impact: Melts ice-caps leading to rising sea levels and coastal flooding; alters climatic patterns causing more rainfall in tropical regions; causes large-scale ecosystem destruction and affects agricultural plant life.
- Mitigation: Reducing the use of fossil fuels by shifting to LNG, LPG, and CNG; adopting renewable energy (solar, wind, biofuels, biogas); promoting afforestation to absorb carbon dioxide; and checking deforestation.
Ozone Depletion
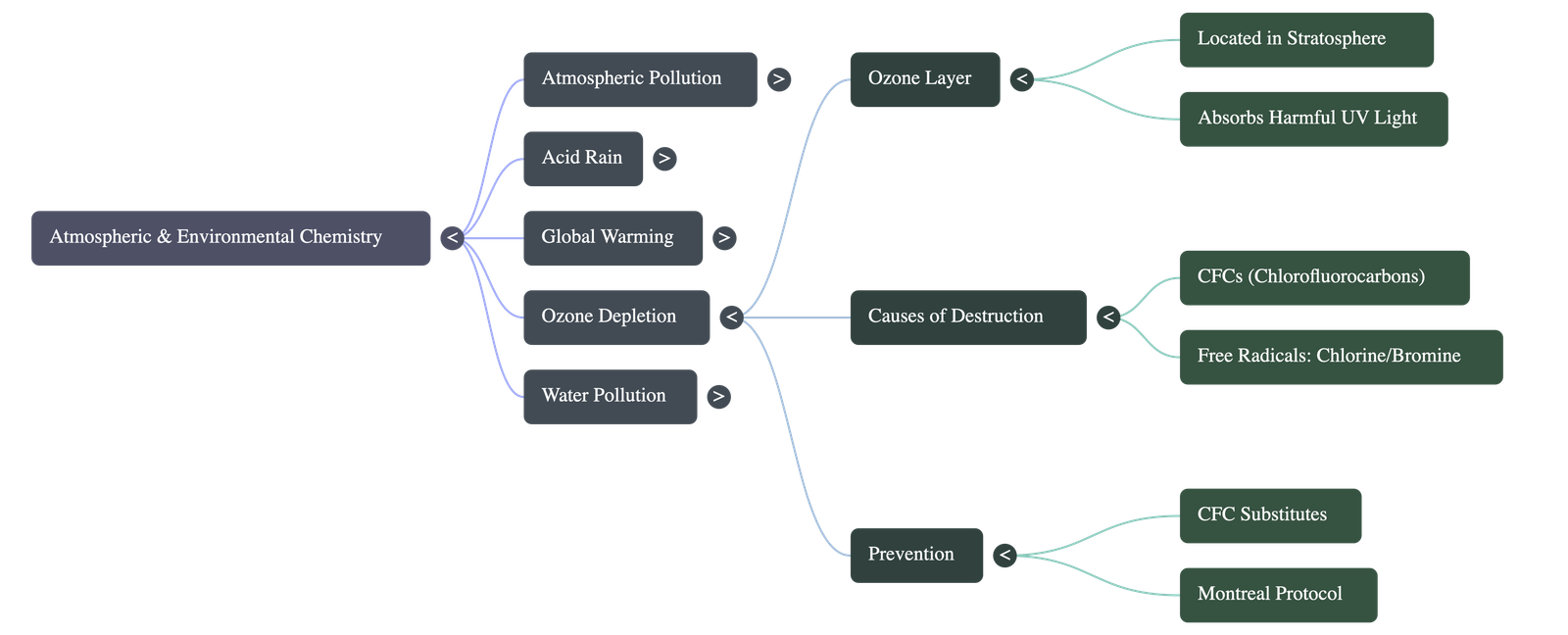
- The Ozone Layer: A layer situated in the stratosphere (about 20-30 kms above the earth) with a high concentration of ozone gas. It acts as a shield protecting life on earth.
- Crucial Function: It absorbs 97-99% of the sun's high-frequency, harmful ultraviolet (U.V.) radiations. U.V. exposure can cause skin cancer, eye cataracts, DNA disruption in humans, and inhibit plant growth and pollen germination.
- Formation & Balance: Highly energetic U.V. radiation breaks down oxygen molecules into atomic oxygen, which then combines with molecular oxygen to form ozone. A natural cycle maintains the balance of ozone produced and destroyed.
- Causes of Depletion: The natural balance is disrupted by man-made chemicals, primarily Chlorofluorocarbons (CFCs), which account for about 80% of ozone depletion. Other chemicals include methyl chloride, methyl bromide, and carbon tetrachloride. U.V. rays break these down, releasing free radicals like active chlorine atoms that catalyze the breakdown of ozone molecules.
- Prevention: Substituting CFCs with safer alternatives (like HCFCs), initiating CFC-free product labeling, and enforcing international treaties like the Montreal Protocol to phase out ozone-depleting substances.
Water Pollution
- Definition: The contamination of water bodies by various pollutants such as domestic sewage, industrial wastes, viruses, and bacteria.
- Beneficial Substances in Water: Dissolved oxygen is vital for aquatic life. Trace minerals like calcium (for bones and teeth), potassium (regulates fluid balance), zinc (immunity), and copper (brain function) are beneficial for human health.
- Harmful Contaminants: Toxic heavy metals (mercury, lead, cadmium), industrial chemical wastes (benzene), polymers (plastics), agricultural runoff (pesticides, nitrates), and biological pathogens (bacteria and viruses).
- Health Hazards: These contaminants cause illnesses like severe diarrhea, endocrine malfunction, and organ damage.
- Minamata Disease: A severe neurological condition first discovered in Japan, caused by the consumption of fish heavily contaminated with mercury discharged from industrial waste. It leads to severe sensory disturbances, lack of coordination, and impairments in speech and hearing.
Quick Navigation:
| | | |
1 / 1
Quick Navigation:
| | | |