Quick Navigation:
| | | |
The Periodic Table
1. Introduction to Elements & Classification
- Elements: The basic unit of matter, comprising only one kind of atom. Currently, around 118 elements have been discovered.
- Need for Classification: To efficiently study the properties of elements, they need to be grouped systematically so that elements with maximum resemblances are placed together while isolating those with different properties.
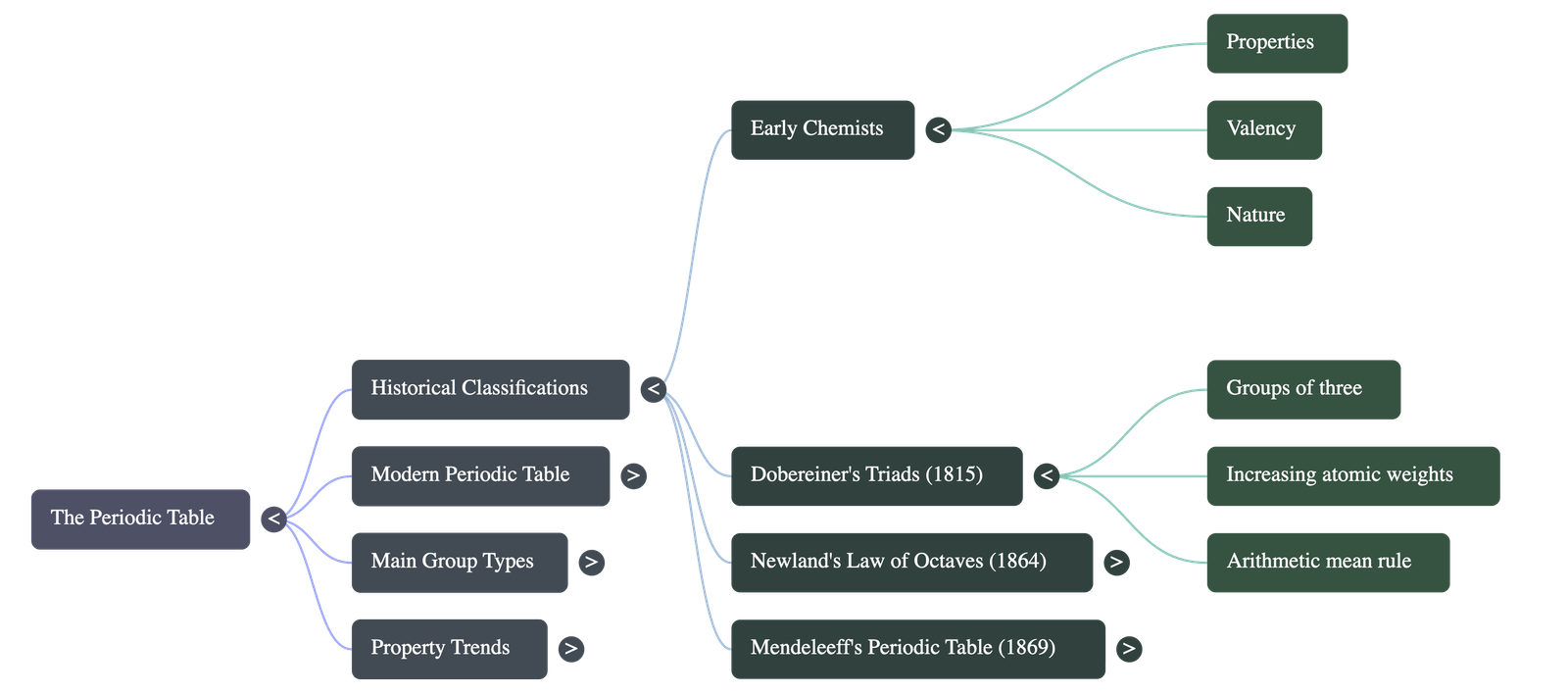
- Early Chemists' Approach: Initially attempted to classify elements based on properties, valencies, and metallic/non-metallic nature. This was discarded because elements often show variable valency and dual (both metallic and non-metallic) behavior.
2. Historical Attempts at Classification
- Dobereiner's Triads (1815):
- Arranged chemically analogous elements in increasing order of atomic weights into distinct groups of three called "triads".
- Law: The atomic weight of the middle element was found to be generally the average (arithmetic mean) of the other two elements (e.g., Lithium, Sodium, Potassium).
- Limitations: This method only produced a disordered collection and did not hold true for all known elements.
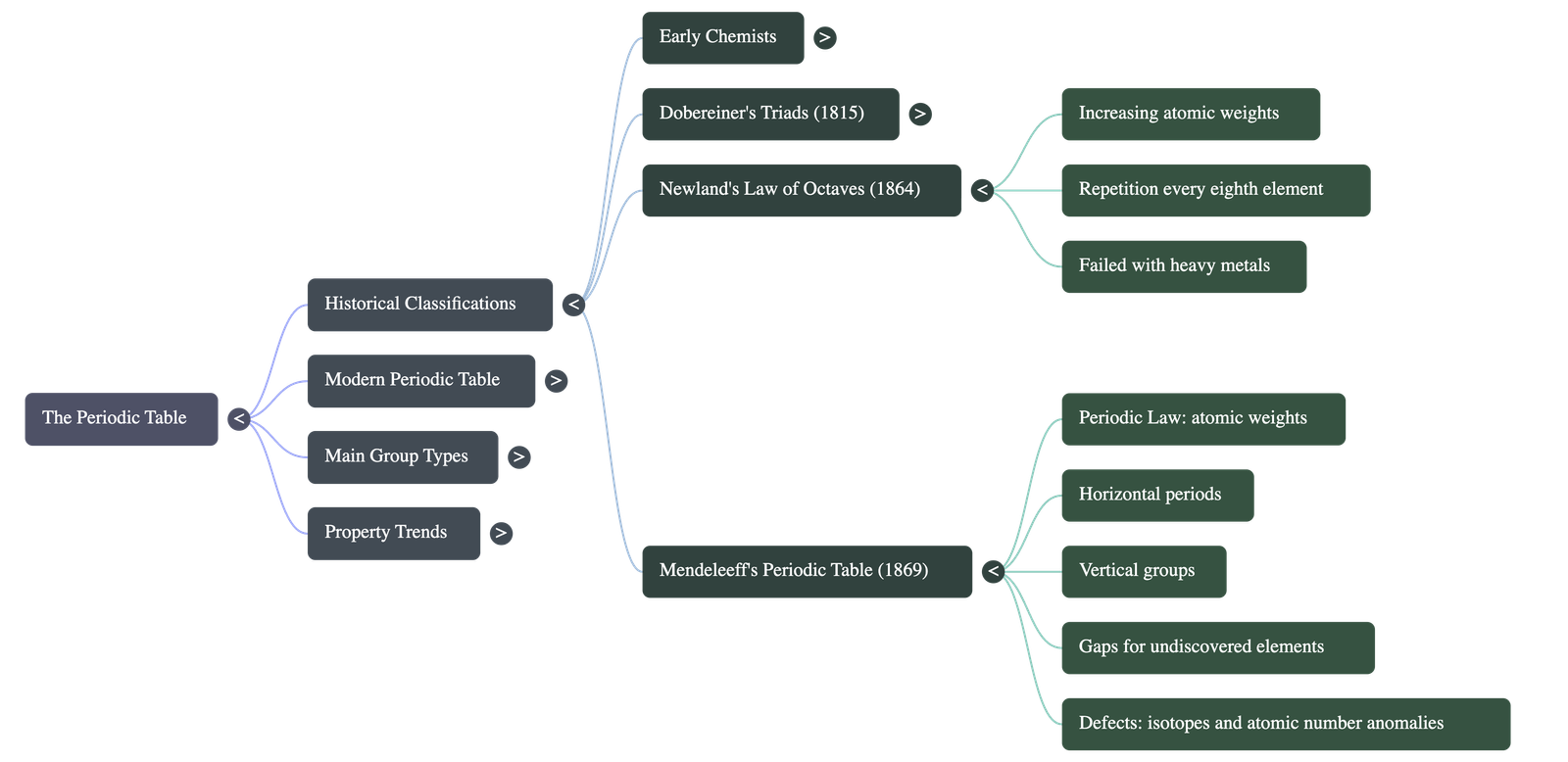
- Newland's Law of Octaves (1864):
- Arranged elements in series of eight in increasing order of atomic weights.
- Law: Every eighth element showed a repetition of physical and chemical properties of the first element, similar to notes on a musical scale.
- Limitations: It successfully grouped lighter metals (like Li and Na) but failed with heavy metals beyond Potassium. It also failed to leave blank spaces for undiscovered elements.
3. Mendeleeff's Contributions (1869)
- Mendeleeff's Periodic Law: The physical and chemical properties of elements are periodic functions of their atomic weights.
- Structure: Elements were arranged in a table consisting of horizontal rows called "periods" and vertical columns called "groups". Elements in the same group had the same valency and similar chemical properties.
- Key Merits:
- Predicted the existence and properties of undiscovered elements by leaving strategic gaps (e.g., eka-boron, eka-aluminium, eka-silicon).
- Corrected the incorrect atomic weights of certain elements using the weights of adjacent elements.
- Defects in Mendeleeff's Table:
- Anomalous Pairs: Some elements with higher atomic weights were placed before elements with lower atomic weights (e.g., Cobalt was placed before Nickel).
- Position Issues: Rare earths, actinides, and isotopes lacked a justifiable position when arranged strictly by atomic weight.
4. The Modern Periodic Law & Table (1912)
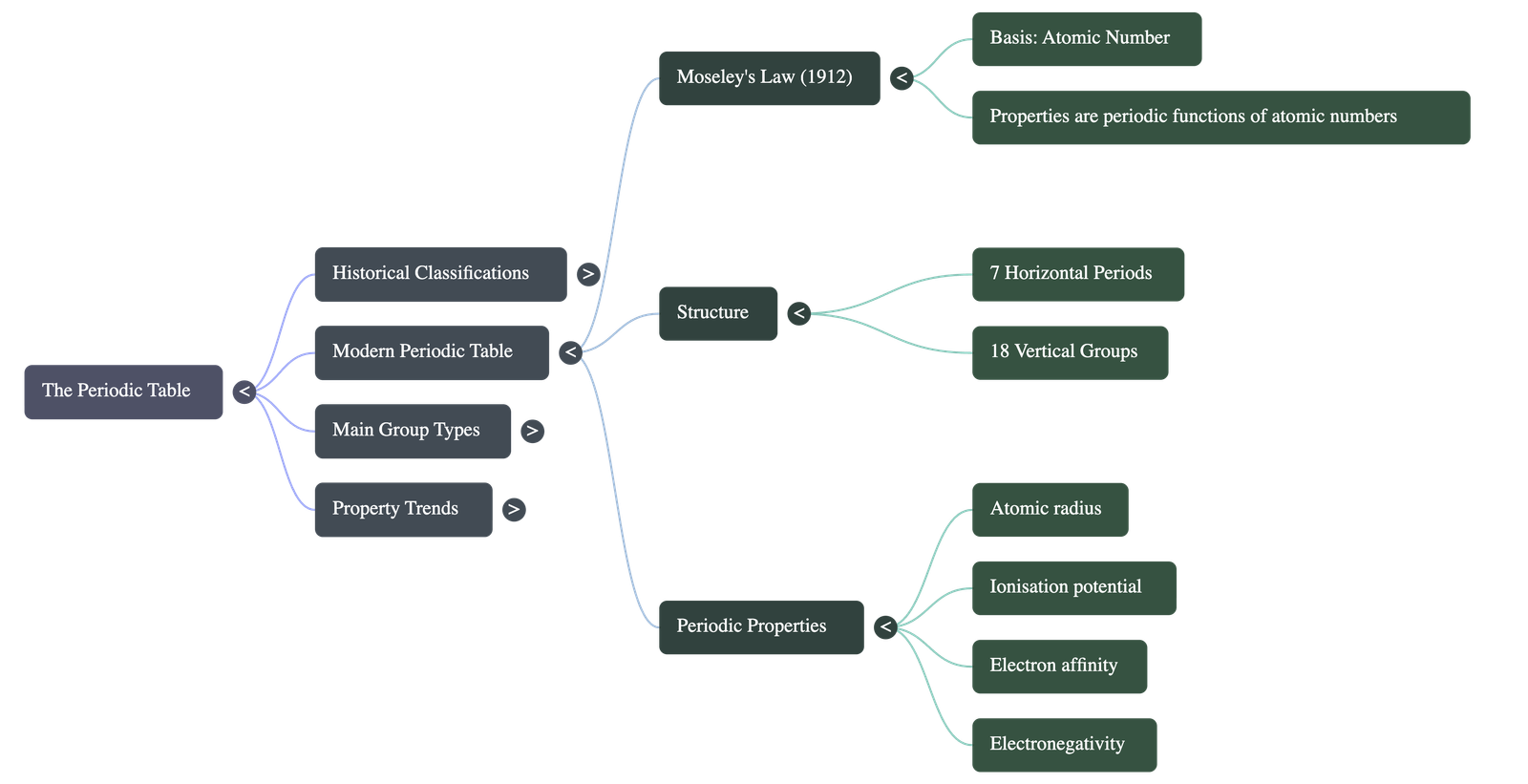
- Moseley's Classification: Solved Mendeleeff's defects by changing the basis of classification from atomic weights to atomic numbers.
- Modern Periodic Law: The physical and chemical properties of elements are periodic functions of their atomic numbers.
- Reason for Periodicity: After definite intervals of atomic number, elements exhibit a recurrence of similar electronic configurations in their valence shell, leading to similar properties.
5. Salient Features of the Modern Periodic Table
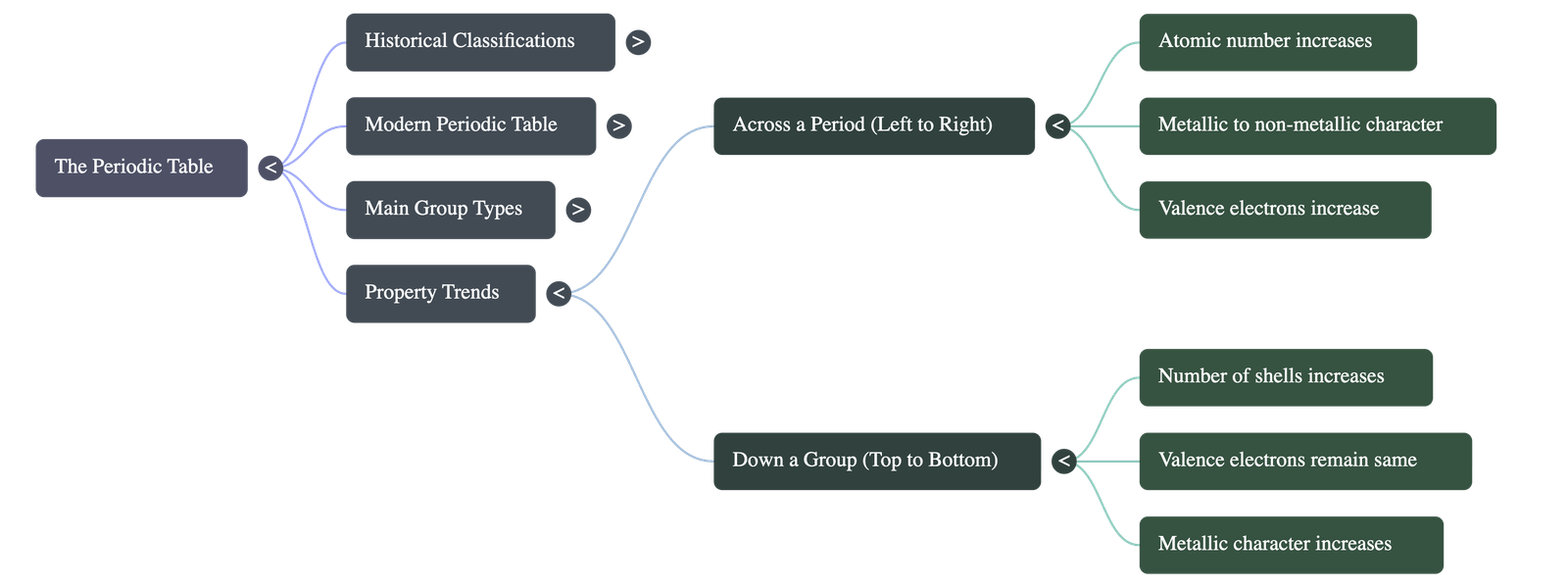
- Periods (7 Horizontal Rows):
- The period number signifies the number of electron shells an element possesses (e.g., Period 1 has 1 shell; Period 3 has 3 shells).
- Period 1: Shortest period containing 2 elements (H, He).
- Periods 2 & 3: Short periods containing 8 elements each.
- Trends Across a Period: As you move left to right, valence electrons increase by one, while the number of electron shells remains the same. A clear transition from metallic to non-metallic character is observed.
- Groups (18 Vertical Columns):
- The group number correlates with the number of valence electrons. Elements in the same group have similar or graded properties and the same valency.
- Trends Down a Group: The number of valence electrons remains the same, but the total number of electron shells increases by one with each step. Metallic character progressively increases down a group.
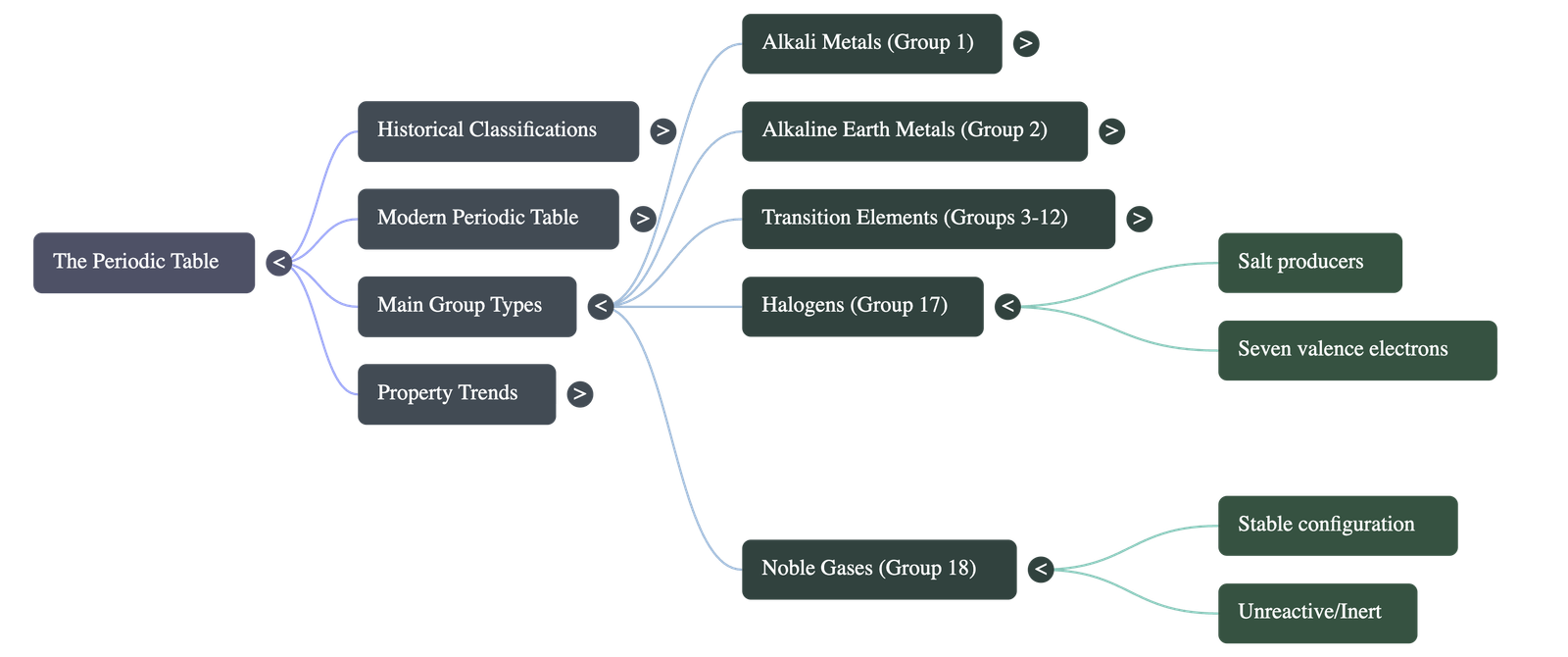
6. Specific Element Families (Groups)
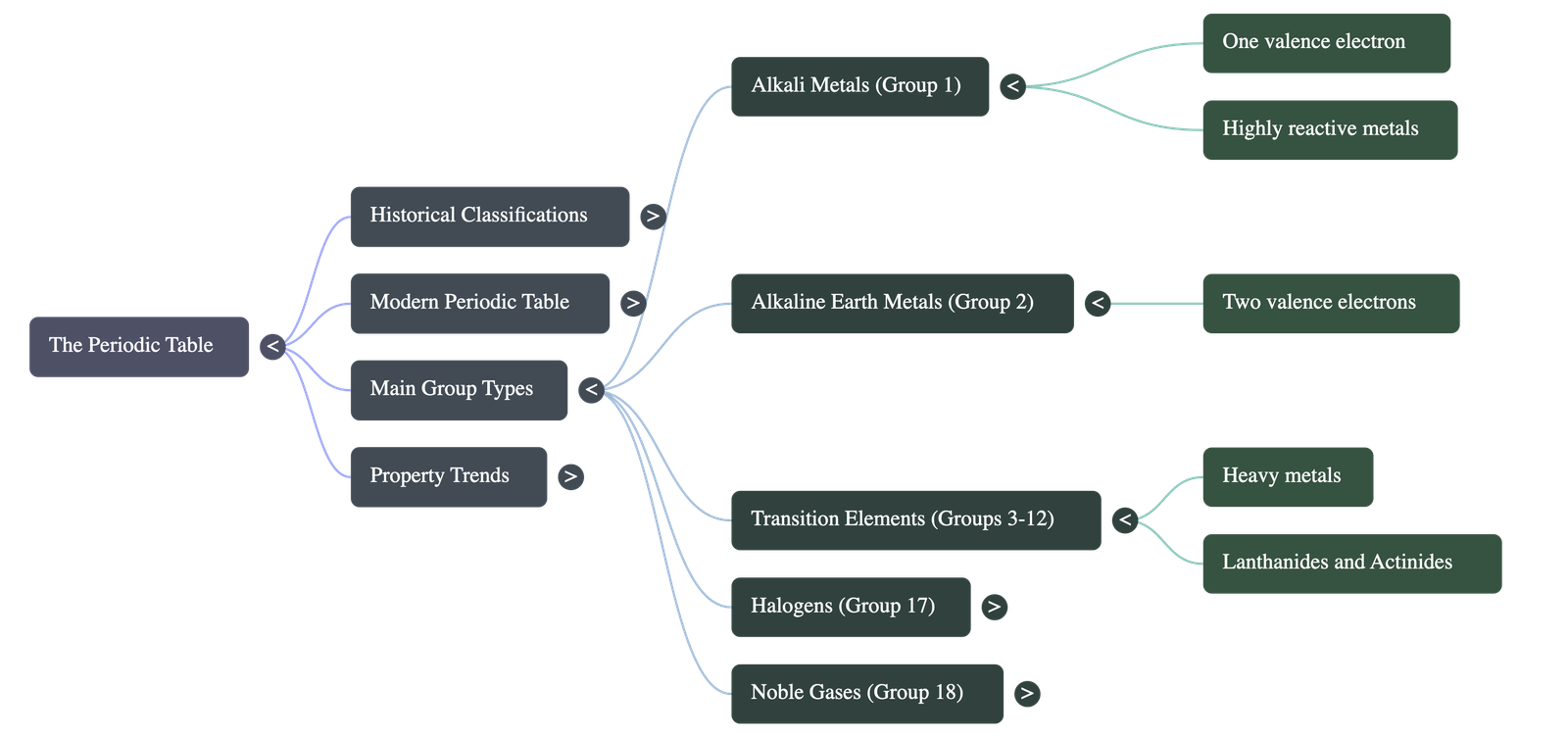
- Group 1 - Alkali Metals: (Li, Na, K, Rb, Cs, Fr)
- Highly reactive, light, electropositive metals.
- They have one valence electron, act as strong reducing agents (electron donors), and react with water to form strong alkalis.
- Group 2 - Alkaline Earth Metals: (Be, Mg, Ca, Sr, Ba, Ra)
- Light metals with two valence electrons. Their properties are similar to Alkali metals but they are slightly less reactive.
- Groups 3 to 12 - Transition Elements:
- Heavy metals placed in the center of the periodic table.
- Inner Transition Elements: Lanthanides (Rare earths) and Actinides (Radioactive elements) are placed at the bottom in two horizontal rows.
- Group 17 - Halogens: (F, Cl, Br, I)
- Highly reactive, electronegative non-metals.
- They have seven valence electrons, are strong oxidizing agents (electron acceptors), and act as "salt producers" when reacting with metals. Fluorine is the most reactive.
- Group 18 - Noble/Inert Gases: (He, Ne, Ar, Kr, Xe, Rn)
- Stable electronic configuration with an outermost shell completely filled (zero valency).
- They are unreactive, monoatomic gases.
7. Key Group Characteristics & Trends
- Electronegativity: The tendency of an atom to attract electrons towards itself.
- Increases across a period (left to right) and decreases down a group.
- Non-metals gain electrons and are electronegative elements.
- Metals lose electrons and are electropositive elements.
- Allotropy: The ability of an element to exist in more than one physical form. This is largely exhibited by elements in Groups 14, 15, and 16.
- Bridge Elements: Elements of the second period that show a resemblance in properties with elements of the third period in the next group, demonstrating a "diagonal relationship" (e.g., Lithium & Magnesium; Beryllium & Aluminum).
- Segregation of Elements: The modern table successfully separates metals (extreme left and center), non-metals (upper right corner), and noble gases (extreme right).
Quick Navigation:
| | | |
1 / 1
Quick Navigation:
| | | |