Quick Navigation:
| | | |
Study of the First Element - Hydrogen
1. Position of Hydrogen in the Periodic Table
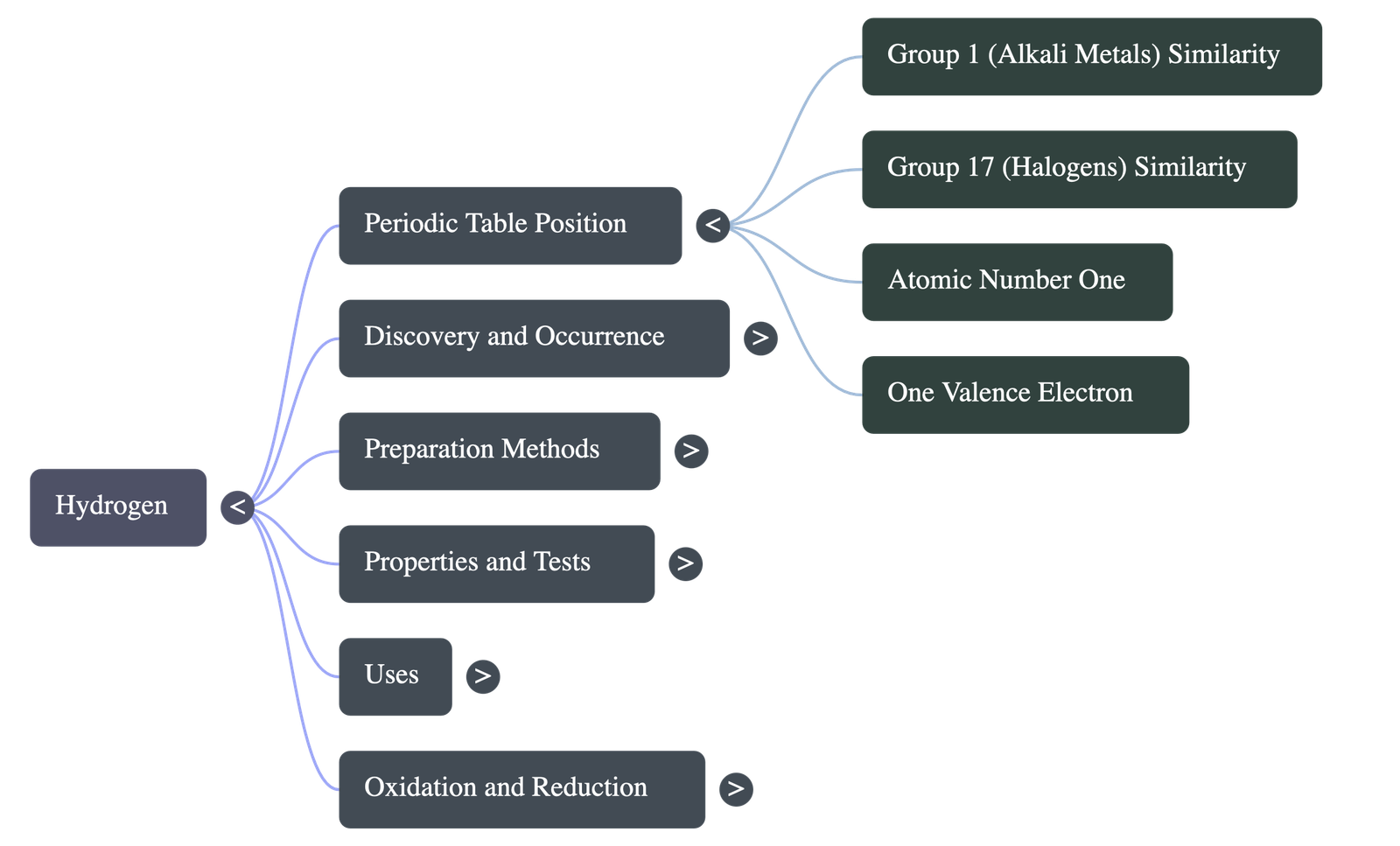
- Atomic Properties: Atomic number is 1, and it has 1 valence electron. It is the first element of the periodic table.
- Dual Nature: Hydrogen has the simplest electronic configuration and exhibits a dual nature, sharing similarities with both Group 1 (Alkali Metals) and Group 17 (Halogens).
- Similarities with Alkali Metals: Exhibits electropositive character (loses 1 electron to form positive ions), has a valency of one, has a strong affinity for non-metals, and acts as a reducing agent.
- Similarities with Halogens: Requires one electron to achieve the nearest noble gas configuration, exhibits electronegative character, shows both electrovalency and covalency, and exists as a diatomic molecule.
2. Discovery & Occurrence
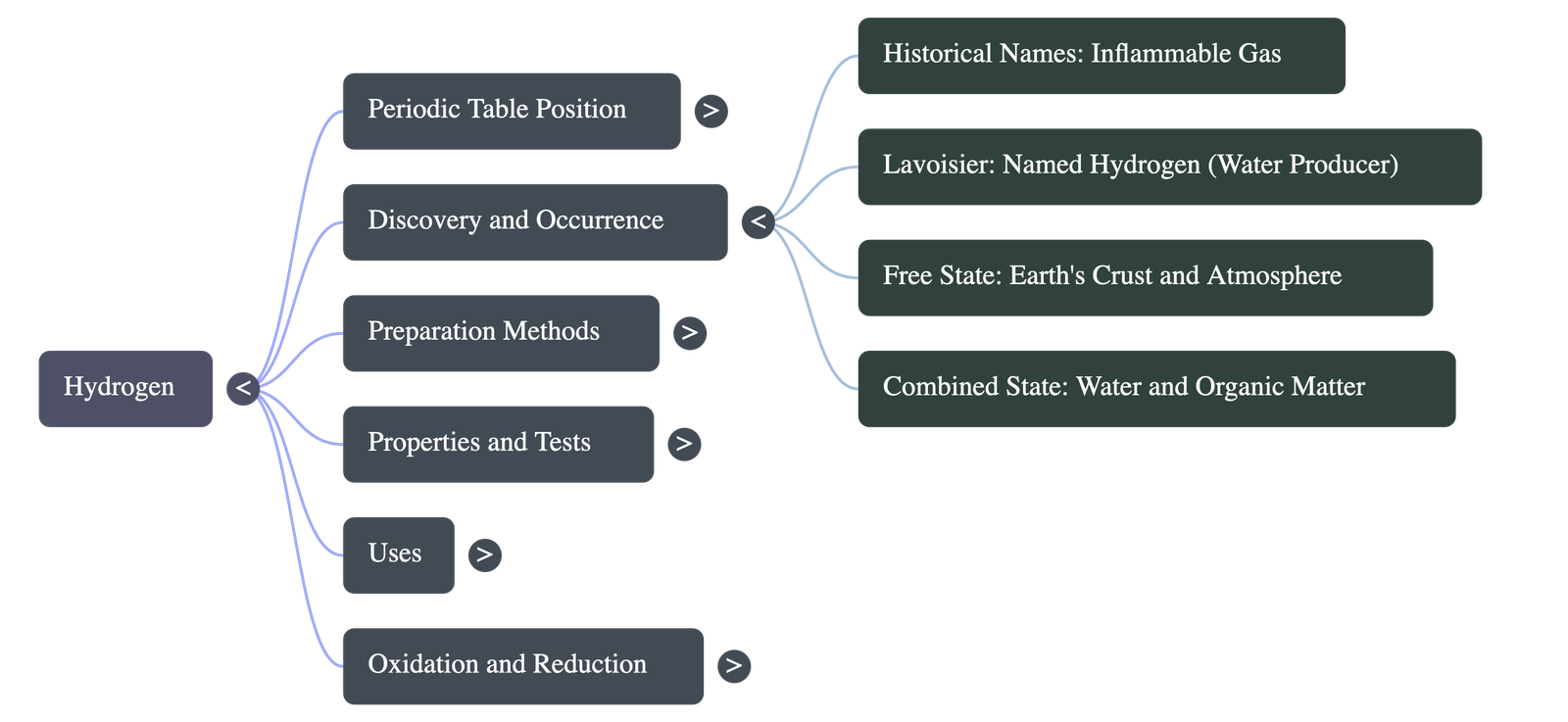
- Discovery: Initially called 'inflammable gas' by Robert Boyle. Henry Cavendish (1776) first prepared it in a pure state and recognized it as an element. Lavoisier (1783) gave it the name "hydrogen" meaning "water producer".
- Free State: Makes up roughly 1% of the Earth's crust and is seldom found free on Earth, but is found in volcanic gases and extensively around the sun and stars.
- Combined State: Very abundant in combined forms such as water, plant and animal tissues, and most organic substances like natural gas, petroleum, proteins, and carbohydrates.
3. Preparation of Hydrogen - General Methods
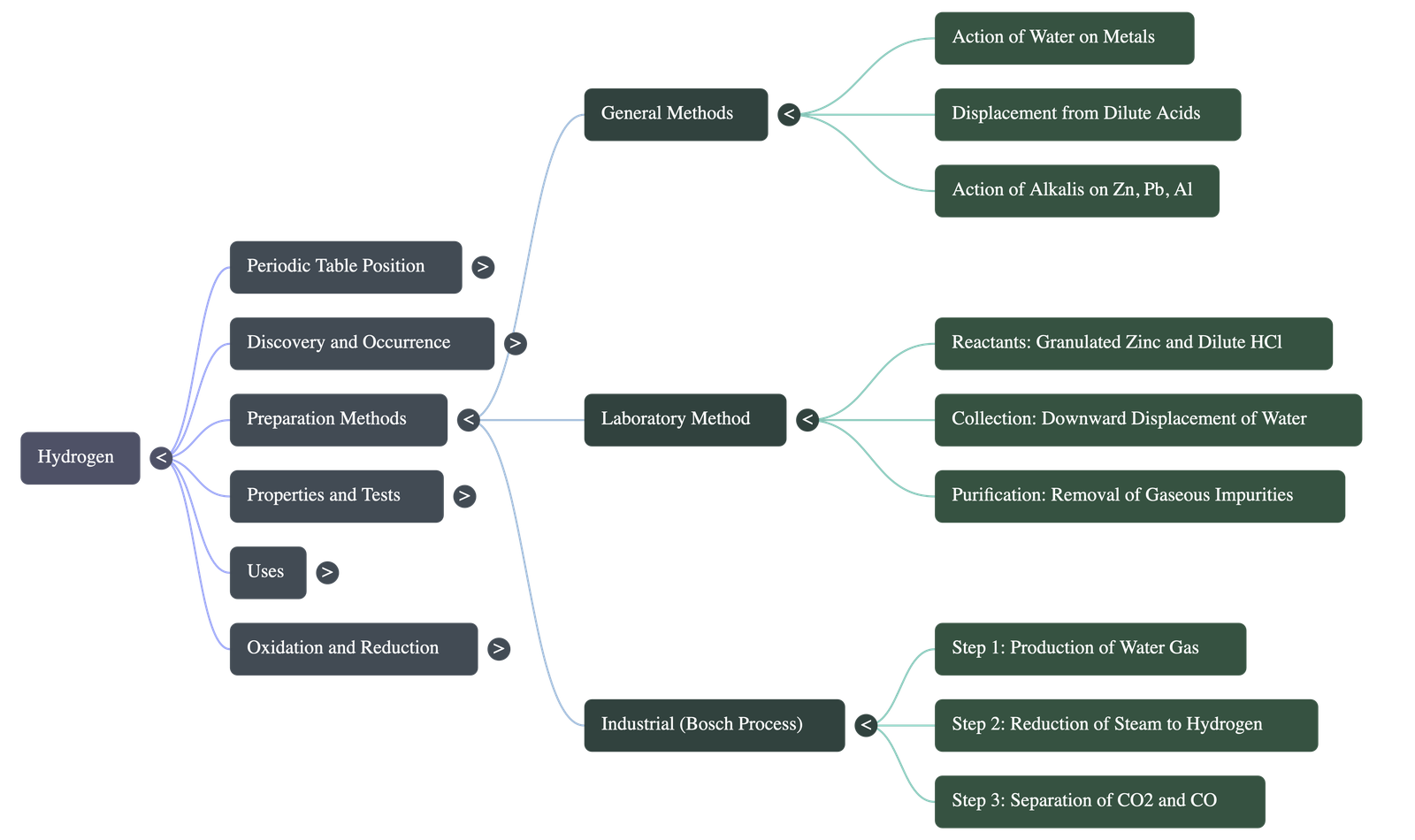
- From Water:
- Potassium, Sodium, and Calcium react with cold water to release hydrogen. (Reaction with K and Na is violent and exothermic).
- Magnesium, Aluminium, Zinc, and Iron react with boiling water or steam to form corresponding oxides and hydrogen. The reaction with iron is reversible.
- From Dilute Acids: Active metals (Mg, Al, Zn, Fe) react with dilute hydrochloric acid or sulphuric acid to yield hydrogen. Nitric acid is not used because it is a powerful oxidizing agent that converts the formed hydrogen into water.
- From Alkalis: Metals like Zinc, Lead, and Aluminium have a unique amphoteric nature, allowing them to react with hot concentrated alkalis (NaOH, KOH) to form hydrogen and complex salts like zincates or aluminates.
4. Laboratory Preparation of Hydrogen
- Reactants: Prepared using granulated zinc (which contains trace impurities that act as a catalyst) and dilute hydrochloric or sulphuric acid.
- Apparatus Precautions: The apparatus must be airtight. A naked flame should not be brought near it because hydrogen forms an explosive mixture with air. The thistle funnel must dip below the acid level to prevent gas leakage.
- Collection: Collected by the downward displacement of water because it is nearly insoluble in water. It is not collected over air because it is lighter than air and highly explosive when mixed with it.
- Purification: The evolved gas is passed through multiple solutions (like silver nitrate, lead nitrate, and KOH) and a drying agent (fused calcium chloride) to remove gaseous impurities like arsine, phosphine, hydrogen sulphide, carbon dioxide, and moisture.
5. Industrial Manufacture - Bosch Process
- Step 1 (Production of Water Gas): Steam is passed over white-hot coke at around 1000°C to produce water gas (Carbon monoxide + Hydrogen).
- Step 2 (Reduction of Steam): Water gas is mixed with excess steam and passed over Iron (III) oxide (catalyst) and chromic oxide (promoter) at 450°C. This converts CO into CO2, yielding a further volume of hydrogen.
- Step 3 (Separation):
- Carbon dioxide is removed by dissolving the mixture in water under high pressure or using a caustic potash solution.
- Unreacted carbon monoxide is removed by dissolving it in an ammoniacal cuprous chloride solution.
- Other Methods: Electrolysis of acidified water, electrolysis of brine, and extraction from natural gas.
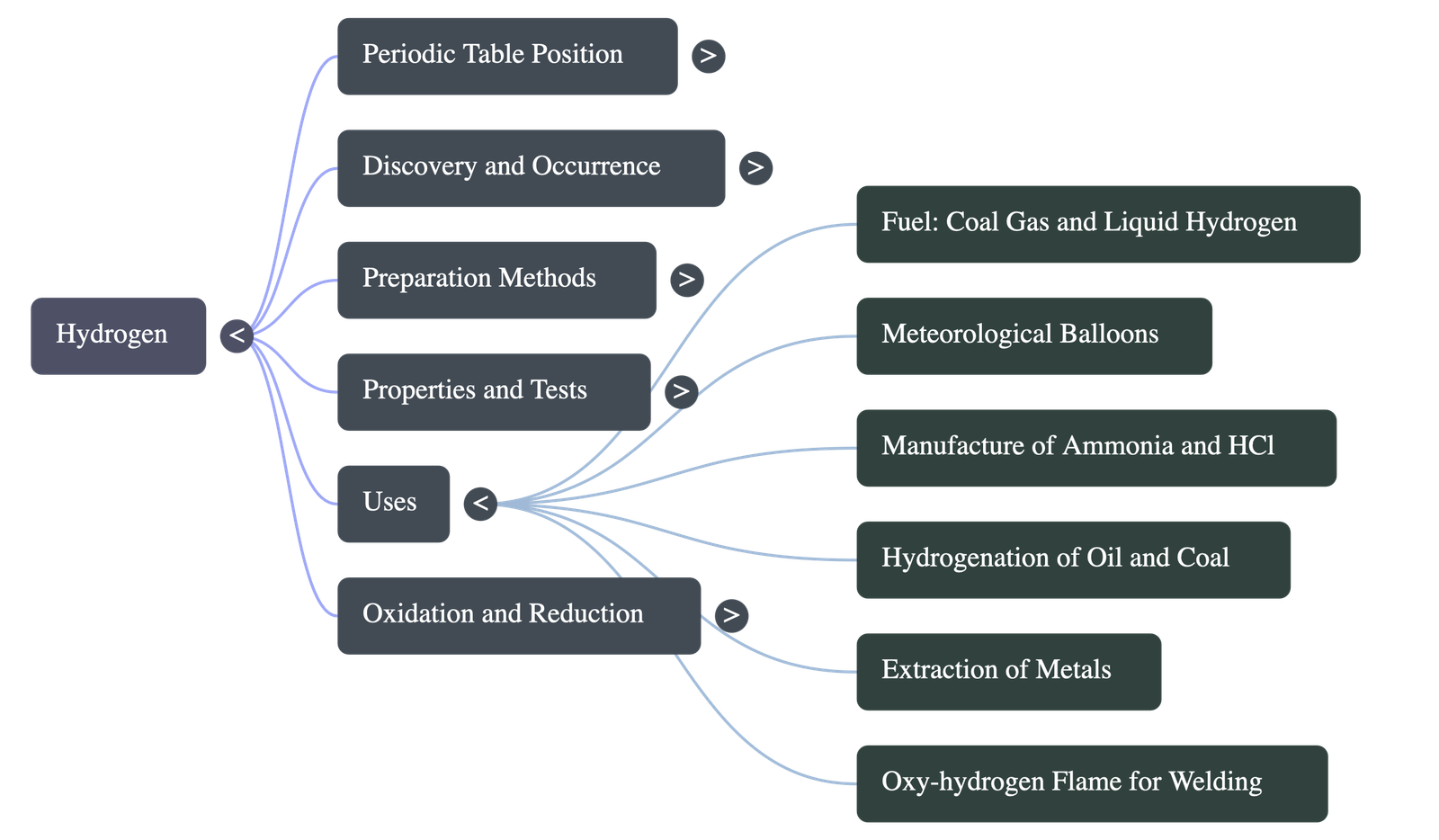
6. Tests & Uses of Hydrogen
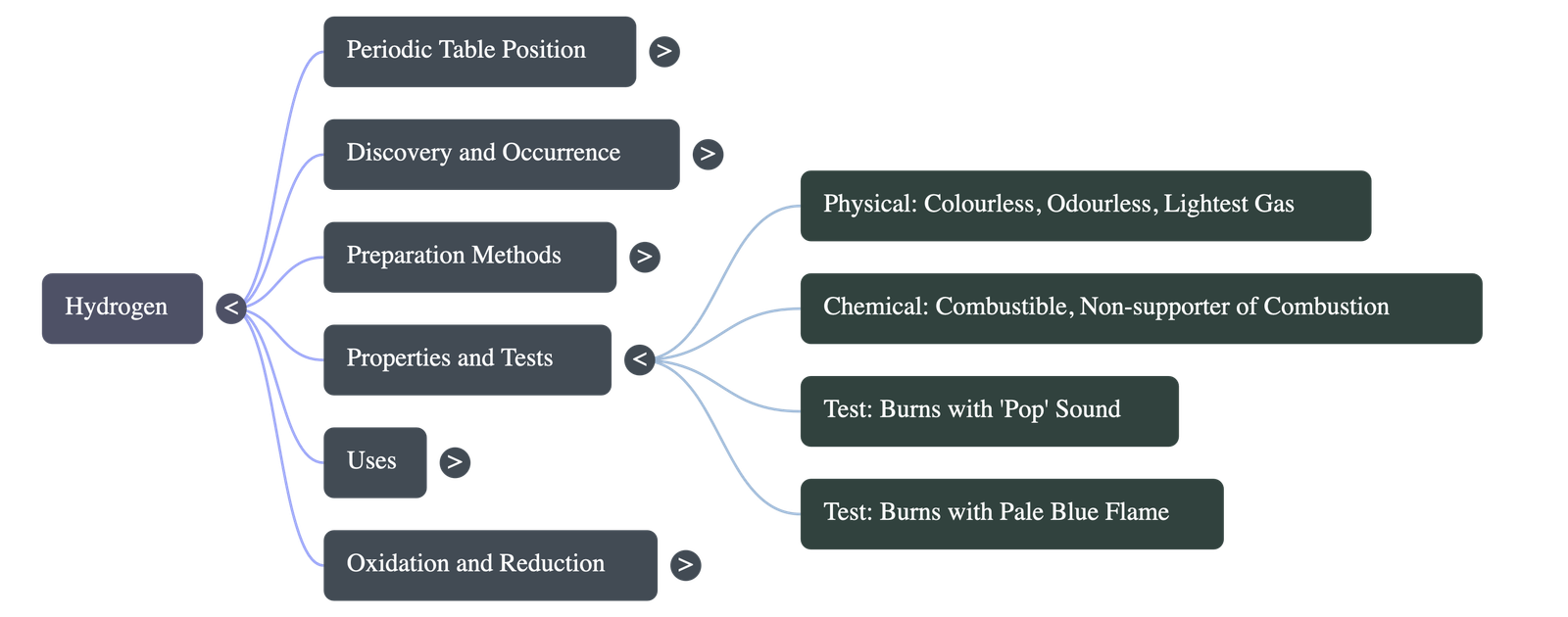
- Tests: Pure hydrogen burns quietly in air with a pale blue flame to form water. A hydrogen-air mixture burns with a characteristic 'pop sound' due to a mild explosion on ignition.
- Uses:
- Fuel: Used in coal gas, water gas, and as a non-polluting liquid fuel.
- Meteorological Balloons: Used due to being lighter than air, though heavily replaced by helium for safety.
- Industrial Manufacturing: Essential for producing Ammonia (Haber process) and Hydrogen chloride.
- Hydrogenation: Used to convert vegetable oils into semi-solid fats (hydrogenation of oil) and coal into petroleum-like products.
- Metallurgy: Acts as a reducing agent in the extraction of less active metals (like zinc, iron, copper) from their oxides.
- Welding & Cutting: An oxy-hydrogen flame generates extremely high temperatures (around 2800°C) ideal for welding metals.
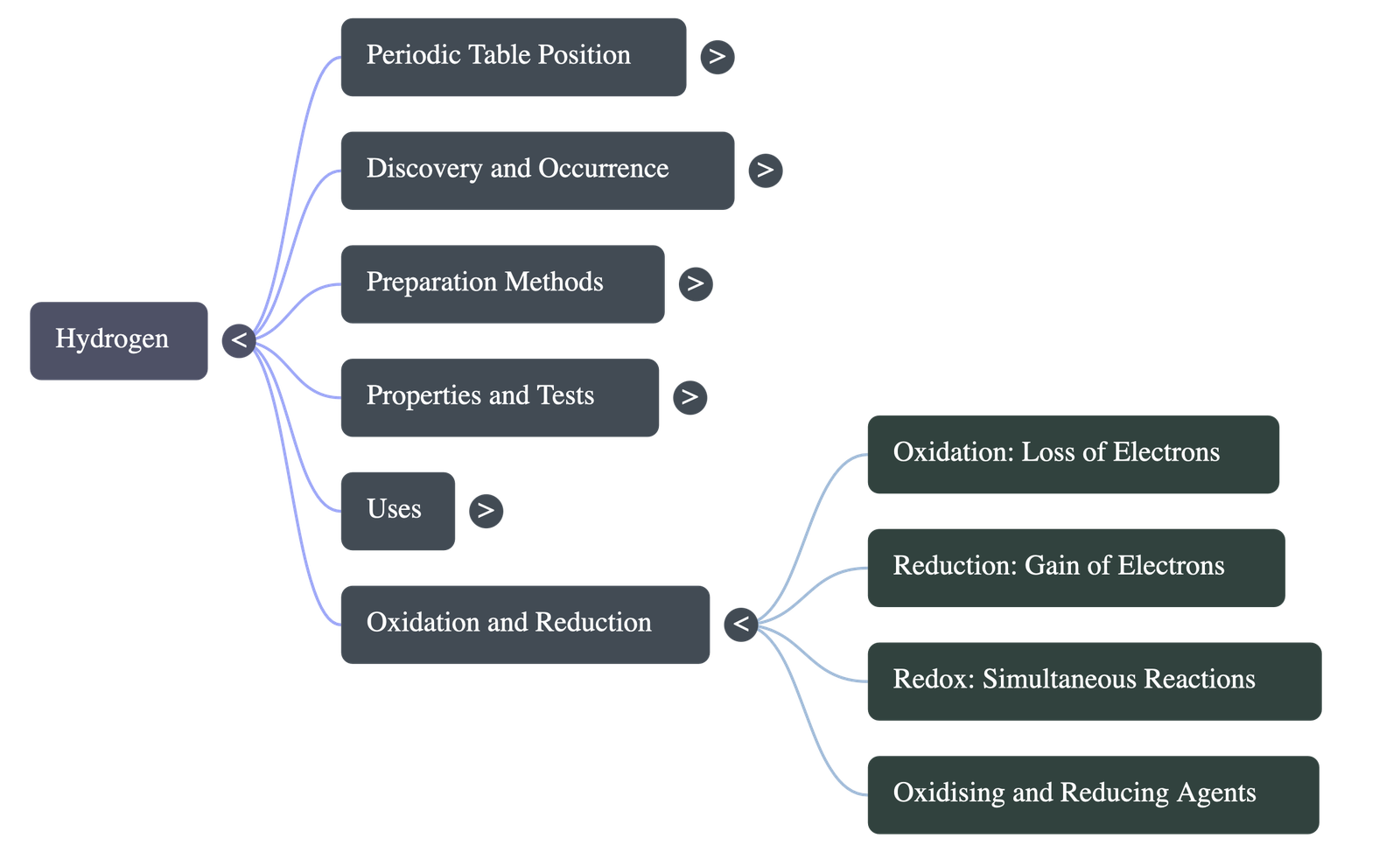
7. Oxidation & Reduction Reactions
- Oxidation involves: Addition of oxygen, addition of an electronegative element, removal of hydrogen, removal of an electropositive element, or the loss of electrons from an atom or ion.
- Reduction involves: Removal of oxygen, removal of an electronegative element, addition of hydrogen, addition of an electropositive element, or the gain of electrons from an atom or ion.
- Redox Reactions: Chemical reactions where oxidation of one substance and reduction of another occur simultaneously.
- Agents:
- Oxidizing Agents: Substances that lose oxygen, gain hydrogen, or accept electrons.
- Reducing Agents: Substances that gain oxygen, lose hydrogen, or donate electrons.
Quick Navigation:
| | | |
1 / 1
Quick Navigation:
| | | |